Researchers Develop New Nanomaterials to Deliver Anticancer Drugs to Kill Cancer Cells
Researchers at UCLA have successfully manipulated nanomaterials to create a new drug-delivery system that promises to solve the challenge of the poor water solubility of today’s most promising anticancer drugs and thereby increase their effectiveness.
The poor solubility of anticancer drugs is one of the major problems in cancer therapy because the drugs require the addition of solvents in order to be easily absorbed into cancer cells. Unfortunately, these solvents not only dilute the potency of the drugs but create toxicity as well.
In a paper scheduled to be published in the nanoscience journal Small in June, researchers from UCLA’s California NanoSystems Institute and Jonsson Cancer Center report a novel approach using silica-based nanoparticles to deliver the anticancer drug camptothecin and other water-insoluble drugs into human cancer cells.
The study is led by Fuyu Tamanoi, UCLA professor of microbiology, immunology and molecular genetics and director of the Jonsson Cancer Center’s Signal Transduction and Therapeutics Program Area, and Jeffrey Zink, UCLA professor of chemistry and biochemistry.
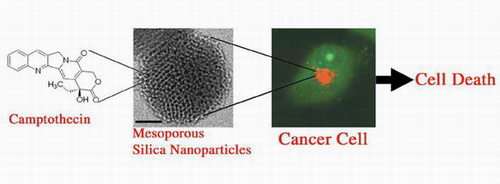
Tamanoi and Zink devised a method for incorporating the representative hydrophobic anticancer drug camptothecin into the pores of fluorescent mesoporous silica nanoparticles and delivering the particles into a variety of human cancer cells to induce cell death. The results suggest that the mesoporous silica nanoparticles might be used as a vehicle to overcome the insolubility problem of many anticancer drugs.
“Silica nanomaterials show promise for delivering camptothecin and other water-insoluble drugs,” Tamanoi said. “We have successfully loaded hydrophobic anticancer drugs into mesoporous nanoparticles and delivered them into human cancer cells to induce cell death.”
“The beauty of our findings is that these nanoparticles are biocompatible, contain tubular pores and are relatively easy to modify,” Zink said. “Additional modification by attaching a ligand against a cancer-cell-specific receptor can make the nanoparticles recognizable by cancer cells.”
A critical obstacle and challenge for cancer therapy is the limited availability of effective biocompatible delivery systems. Since many effective anticancer agents have poor water solubility, the development of novel delivery systems for these molecules without the use of organic solvents has received significant attention.
Camptothecin (CPT) and its derivatives are considered to be among the most effective anticancer drugs of the 21st century. Although studies have demonstrated their effectiveness against carcinomas of the stomach, colon, neck and bladder, as well as against breast cancer, small-cell lung cancer and leukemia in vitro, clinical application of CPT in humans has only been carried out with CPT derivatives that have improved water solubility.
“In order to be used on humans, current cancer therapies such as CPT or Taxol, which are poorly water soluble, must be mixed with organic solvents in order to be delivered into the body,” Tamanoi said. “These elements produce toxic side effects and in fact decrease the potency of the cancer therapy.”
To overcome these problems, drug delivery systems using pegylated polymers, liposomal particles or albumin-based nanoparticles have been developed.
The new research findings show that mesoporous silica nanoparticles offer great potential and a promising approach to the delivery of therapeutic agents into targeted organs or cells. The pores in the nanoparticles could be closed by constructing an appropriate cap structure. This provides the ability to control the release of anticancer drugs by external stimuli.
Source: University of California - Los Angeles