New Law for Resolution Allows Unprecedented Sharpness in Fluorescence Microscopy

Researchers from the Max Planck Institute for biophysical Chemistry in Göttingen establish a new law allowing unlimited optical resolution in fluorescence microscopy
Max Planck researchers have succeeded in overcoming the law postulated by Ernst Abbe in 1873 for diffraction limited resolution in light microscopes. Stefan Hell and his co-workers have established a new law that promises unlimited resolution in fluorescence microscopy. Future applications range from the imaging of cell interiors to the measuring of lithographic structures in microchip manufacturing, and substantial improvements in the quantification of the reaction kinetics of organic molecules (Phys. Rev. Lett., April 15 and Phys. Rev. Lett. May 6).
Images with resolution at and beyond the diffraction limit shown in the left and right column, respectively. Upper row: Pores in a porous membrane marked with a fluorescent dye shown with normal resolution cannot be discerned as such (a). The same imaging carried out parallel with STED microscopy clearly brings out the structure to light (b). The term confocal indicates that the reference images (a,c) were recorded in the confocal microscopy imaging mode which currently is the best resolving diffraction limited fluorescence microscopy method. Lower row: Fluorescence dye marked nanostructures produced by electron beam lithography in a polymer shown with normal resolution (c) and then using STED (d). The raw data in (c) and (d) after imaging was subjected to linear deconvolution to mathematically slightly enhance the resolution. In spite of this, the image in (c) does not show the structures, whereas the images with the STED microscope can resolve lines of 80 nm width and 40nm separation between the lines (d). Thus, the optical imaging is moving into domains that were until now reserved for the electron microscope. Image: MPI for Biophysical Chemistry
Ever since its invention in the 17th century, the light microscope has served as a key instrument to the advancement of knowledge. For biology, this statement is true even today, since focused light is the only way to examine living cells non-invasively. However, because of the wave nature of light, focused light is subject to diffraction. As early as 1873, Ernst Abbe recognized that this fact imposes an absolute limit on resolution in microscopy. Abbe, whose 100th year death anniversary was in February, captured this limit in a formula, which states that objects at distances that are smaller than a certain limit cannot be separated in a light microscope. This resolution limit can be largely ascribed to the fact that light cannot be focused to a smaller spot due to diffraction. It seemed impossible to image details smaller than a distance where delta d is equal to 200nm using conventional lenses and visible light.
For a long time, Abbe’s law was regarded as being insurmountable. For higher resolution, the textbook knowledge was that a complicated and costly electron or scanning tunneling microscope would be required. However, over the past few years, researchers from the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany, have developed with Stimulated Emission Depletion (STED) microscopy, a physically consistent concept for breaking the diffraction limited resolution limit in fluorescence microscopy, and then verified it in experiments. Unlike in the light microscopes conceivable so far, in a STED microscope, the relevant focal fluorescence spot can, in principle, be reduced in size to the size of a molecule (2-5 nm). This is due to the fact that the spot size is no longer subject to Abbe’s formula, but to a new law that differs from Abbe’s original formula in a crucial factor - a square root term [1,2]:
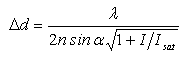
The new law, named after RESOLFT (reversible saturable optical fluorescent transition), which allows unlimited optical resolution in fluorescence microscopy. Image: MPI for biophysical Chemistry / Stefan Hell
In one of their latest publications [2], the Göttingen-based research team verifies this new law with their experiments. They show that even with conventional objectives and focused light, resolutions of 16 nm are feasible. Therefore for the first time ever, it has been demonstrated experimentally that even with focused-light optics, resolutions at the nano-level are possible in fluorescing samples. In an additional publication [3], the researchers show that STED microscopy allows one to image lithographic structures (which had been previously dyed with a fluorescent dye) of width as small as 40-80 nm, a size previously reserved for electron microscopy. This may be of importance for the lithographic manufacturing of microchips.
The reduction in size of the diameter of the effective fluorescence focus also has fundamental implications for methods that use fluorescence fluctuations to study the reaction kinetics of molecules in solutions. The smaller the focal volumes, the more effective and sensitive are these methods. Fluorescence fluctuation methods were also limited by diffraction as a result. Therefore, in a fourth study [4], the research team from Göttingen shows that using STED, measurement volumes notably below the resolution limit can be produced without confining the solution itself mechanically. This method may substantially improve the analysis of pharmaceutical active ingredients and protein interactions in cells in future.
Original work:
[1]Hell, S. W.
Strategy for far-field optical imaging and writing without diffraction limit.
Phys. Lett. A 326(1-2): 140-145 (2004)
[2] Westphal, V., and S. W. Hell
Nanoscale Resolution in the Focal Plane of an Optical Microscope.
Phys. Rev. Lett. 94: 143903 (2005)
[3] Westphal, V., J. Seeger, T. Salditt and S. W. Hell
Stimulated Emission Depletion Microscopy on Lithographic Nanostructures.
J. Phys. B: At. Mol. Opt. Phys. 38: S695 - S705 (2005)
[4] Kastrup, L., H. Blom, C. Eggeling, S. W. Hell
Fluorescence Fluctuation Spectroscopy in Subdiffraction Focal Volumes.
Phys. Rev. Lett. 94: 178104 (2005)
Max Planck Institute for Biophysical Chemistry
Source: Max Planck Society for the Advancement of Science