World's smallest periscopes
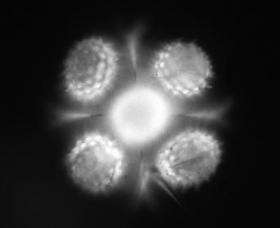
(PhysOrg.com) -- A team of Vanderbilt scientists have invented the world's smallest version of the periscope and are using it to look at cells and other micro-organisms from several sides at once.
"With an off-the-shelf laboratory microscope you only see cells from one side, the top," says team member Chris Janetopoulos, assistant professor of biological sciences. "Not only can we see the tops of cells, we can view their sides as well - something biologists almost never see."
The researchers have dubbed their devices "mirrored pyramidal wells." As the name implies, they consist of pyramidal-shaped cavities molded into silicon whose interior surfaces are coated with a reflective layer of gold or platinum. They are microscopic in dimension - about the width of a human hair - and can be made in a range of sizes to view different-sized objects. When a cell is placed in such a well and viewed with a regular optical microscope, the researcher can see several sides simultaneously.

"This technology is exciting because these mirrored wells can be made at very low cost, unlike other, more complex methods for 3D microscopy," says Assistant Professor of the Practice of Biomedical Engineering Kevin Seale.
According to Ron Reiserer, "This could easily become as ubiquitous as the microscope slide and could replace more expensive methods currently used to position individual cells." Reiserer is a lab manager at the Vanderbilt Institute for Integrative Biosystems Research and Education (VIIBRE) who helped design the protocol used to make the micropyramids.
The Vanderbilt group is not the first to make microscopic pyramidal wells, but it is the first to apply them to make 3D images of microorganisms. In 2006, a group of scientists in England created pyramidal micromirrors and applied them to trapping atoms. And last spring researchers at the National Institute of Standards and Technology used similar structures to track nanoparticles.
The Vanderbilt researchers reported their achievement last September in the Journal of Microscopy. Dmitry A. Markov and Igor Ges, research associates in biomedical engineering; undergraduate researcher Charlie Wright and John P. Wikswo, Gordon A. Cain University Professor and Director of VIIBRE, participated in the development with Janetopoulos, Seale and Reiserer.
So far, the researchers have used the mirrored wells to examine how protozoa swim and cells divide. "The method is particularly well suited for studying dynamic processes within cells because it can follow them in three dimensions," says Janetopoulos. Researchers in his lab have used the wells to track the 3D position of the centrosome - the specialized region of a cell next to the nucleus that is the assembly point where the microscopic polymer tubes that serve as part of the cell's cytoskeleton are assembled before cell division and broken down afterwards.

The mirrored pyramidal wells provide a high resolution, multi-vantage-point form of microscopy that also makes it easier for researchers to measure a number of important cell properties. For his senior thesis, for example, Wright explored how the technique can be used to measure the volume of individual yeast cells with unprecedented accuracy. In addition, Wikswo and Markov plan to create mirrored microchannels to measure how cells are deformed under stress induced by fluid flowing through hair-width channels in order to determine how fluid flow affects cell behavior and attachment.
A popular method for studying biological processes uses genetic engineering to attach genes that produce fluorescent molecules to different cell structures such as specific surface receptors. This procedure makes the targeted cell structures light up when illuminated by ultraviolet light, but strong UV light also has the potential to damage the structures. If the engineered cell structures are put in a micropyramidal well, the fluorescent light that is emitted toward the mirrored sides is reflected upward toward the microscope, allowing the researchers to reduce the intensity of the UV light and its potential for damaging the engineered cells.
According to Janetopoulos, the micropyramids also have a major advantage for single molecule studies. Optical noise is a constant problem when working at the low light levels involved. Being able to pinpoint actual light sources in two or three dimensions allows the researchers to reject spurious signals. This should be useful in quantitative fluorescence or bioluminescence studies: Cells can be genetically modified to glow in the dark to provide a measure of cellular metabolic activity or the expression of a specific gene.
Provided by Vanderbilt University















