February 2, 2009 feature
Shocking: Environmental chemistry affects ferroelectric film polarity the same way electric voltage does
(PhysOrg.com) -- “Ferroelectric materials are interesting scientifically, and, while they are used for some things now, they are potentially useful for even more applications in the future,” Brian Stephenson tells PhysOrg.com. Stephenson is a scientist at Argonne National Laboratory in Argonne, Illinois. He has been working on a project to study chemical switching in a ferroelectric film.
“Normally,” Stephenson continues, “voltage is applied to change the internal structure in ferroelectric materials. You can turn the crystal upside down from the internal point of view. We have shown, I think for the first time, that this can also be done chemically by changing the chemistry of the environment.” The results of the work, which includes scientists from Northern Illinois University and the University of Pennsylvania as well as Argonne, can be found in Physical Review Letters: “Reversible Chemical Switching of a Ferroelectric Film.”
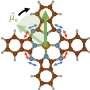
In order to test the process of chemical switching by changing the environment of the ferroelectric film, Stephenson and his colleagues varied the oxygen partial pressure. In situ x-ray scattering was used to “see” the changes in the polarization of the material. The specific ferroelectric material used for the experiment was lead titanate (PbTiO3). The group found that changing the oxygen pressure switched the polarization of the PbTiO3 film in much the same way as the conventional practice of using electrodes and voltage.
The use of x-rays is important, since it allows scientists a peek at what is actually happening inside these materials. “The challenge has been to measure what is going on,” Stephenson admits. “With these thin films, external voltage measurements become more ambiguous. With our x-ray technique, we are able to watch the atomic-scale structure inside these systems.”
“Up until now,” he continues, “we didn’t really think that the environment these ferroelectric materials were in could be just as important as the voltage applied. Fundamentally, we didn’t realize that extra oxygen or missing oxygen at the surface could produce an electric field big enough to affect properties.”
This knowledge will become more important, Stephenson explains, as the demand for smaller devices made from new materials increases. Infrared and terahertz technology, controllable catalysts and chemistry applications on chips represent some of the areas that might benefit from a better knowledge of how switching works with PbTiO3 films.
“Already there are ferroelectric materials used for non-volatile computer memory devices,” Stephenson points out. “But the holy grail of these is a memory element the size of an atom. As films get thinner, understanding the interfacial properties of these materials makes a difference. If the chemistry of the environment can change the polarization, we need to harness this to create new types of devices.”
“The big picture is that we are trying to create new functional materials with interesting properties. We want to understand the way interfaces between different materials work. Ferroelectrics provide a model system where we can produce and measure large effects of the electric fields from the interfaces.”
Additional information: Wang, et. al. “Reversible Chemical Switching of a Ferroelectric Film.” Physical Review Letters (2008). Available online: link.aps.org/doi/10.1103/PhysRevLett.102.047601
Copyright 2009 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.