Using light to move and trap DNA molecules
(PhysOrg.com) -- A major goal of nanotechnology research is to create a "lab on a chip," in which a tiny biological sample would be carried through microscopic channels for processing. This could make possible portable, fast-acting detectors for disease organisms or food-borne pathogens, rapid DNA sequencing and other tests that now take hours or days.
One obstacle has been the difficulty of moving stuff at the nanoscale. Mechanical pumps don't scale down well. So David Erickson, Cornell assistant professor of mechanical and aerospace engineering, and colleague Michal Lipson, Cornell associate professor of electrical and computer engineering, have turned to "optofluidics," using the pressure of light to move and manipulate biological molecules.
Now they have shown that a beam of light can trap and move particles as small as 75 nanometers (nm -- a billionth of a meter) in diameter, including DNA molecules -- some of the smallest material ever manipulated by such a system, the researchers said. Their experiments are described in the Jan. 1, 2009 issue of the journal Nature.
This is possible because of the paradoxical dual nature of light. Light can be thought of as a stream of particles called photons that can exert a force, or as waves of expanding and contracting electric and magnetic fields. If light is confined to a waveguide narrower than its wavelength, the wave overflows and can exert a force beyond the guide. Imagine a nanoscale Indiana Jones chased down a nanotunnel by a photon instead of a granite sphere. if Indy climbs into a tunnel above the one in which the photon is moving, he is still being chased because the photon is bigger than its own tunnel.
Erickson and Lipson first cut microfluidic channels in a chip and placed waveguides directly under the channels. In earlier research published in Optics Express (Oct. 15, 2007), the researchers showed that light in the waveguide could move polystyrene spheres about 3 microns (millionths of a meter) in diameter through the fluid-filled channels. But the "evanescent field" of a light wave that extends beyond the waveguide did not extend far enough or carry enough energy to capture and manipulate smaller biological molecules, they found.
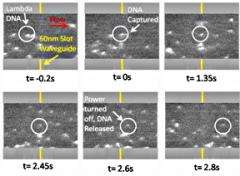
So they turned to a new device created by Lipson: a "slot waveguide" -- two parallel silicon bars 60 nm apart, serving as two parallel wave guides. Light waves traveling along each guide expand beyond its boundaries, but because the parallel guides are so close together, the waves overlap and most of the energy is concentrated in the slot. In addition to creating a more intense beam, this structure allows a beam of light to be channeled through air or water.
As a demonstration, graduate students Allen Yang, Sean Moore, Bradley Schmidt and colleagues in Erickson's and Lipson's research groups laid a slot waveguide across a microscopic fluid channel and showed that light from an infrared laser channeled through the wave guide could trap 75-nm diameter polystyrene spheres and comparably-sized DNA molecules out of a stream of water flowing across it. The light in the slot waveguide extends above the slot and exerts a downward force on a particle entering it, pulling the particle down into the slot. Since light pressure then moves the trapped particles along the slot, such a device could be used to separate biological molecules out of a stream and send them somewhere else for processing, the researchers said. Further development, they added, could make it possible to separate DNA molecules by length for rapid DNA sequencing.
The research was funded by the National Science Foundation, which also supports the Cornell Nanoscale Facility where the devices were manufactured.
Provided by Cornell University





















