Cancer-fighting antibodies

(PhysOrg.com) -- MIT engineers have found that antibodies do not need a particular sugar attachment long believed to be essential to their function, a discovery that could make producing therapeutic antibodies much easier and cheaper in the future.
Therapeutic antibodies are a promising new type of treatment for cancer and other diseases, but their practicality has been limited by the fact that only mammalian cells have the right machinery to build the sugar attachment.
"To date, people have faced limitations in how they were going to make these antibodies because they appeared to require these (sugar) structures," said Dane Wittrup, the C.P. Dubbs Professor of Chemical Engineering, Biological Engineering, member of the Koch Institute for Integrative Cancer Research, and senior author of a paper on the work that appeared in the Dec. 12 online edition of the Proceedings of the National Academy of Sciences.
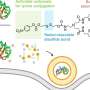
Wittrup and biological engineering graduate student Stephen Sazinsky, co-lead author of the paper, found that antibodies don't need the sugar normally found attached to a certain region of antibody when the sequence is slightly mutated.
Antibodies are a key part of the immune system, roaming around the body to detect invaders such as bacteria and viruses. Each antibody is specific to a particular pathogen. When an antibody finds its target, it first binds to the pathogen, then binds to immune cells, alerting them to attack the pathogen.
The attached sugar was believed necessary to allow antibodies to bind to immune cells, such as NK (natural killer) cells and macrophages. However, the MIT team found that mutant forms of the antibody with no sugar were also able to bind to immune cells.
Knowing this, scientists will now be able to develop therapeutic antibodies that can be mass-produced by bacteria or fungi, a process that is cheaper and faster than using mammalian cells.
There are now dozens of FDA-approved antibody treatments, and hundreds more are in clinical trials. Such therapeutic antibodies can be used to treat cancer, autoimmune diseases, cardiovascular disease and many others.
The new discovery also raises the question of why the sugars are attached to antibodies in the first place. Sazinsky theorizes that the sugar has been evolutionarily conserved as a way for the immune system (and now scientists) to tinker with the binding affinities of different antibodies.
Other authors of the paper are co-lead author Rene Ott of Rockefeller University; Nathaniel Silver, an MIT graduate student in chemistry; Bruce Tidor, MIT professor of biological engineering and computer science; and Jeffrey Ravetch of Rockefeller University.
Provided by MIT