Capturing Sunlight: Indoline Dyes Improve Efficiency of Solar Cells
Solar cell technology is marching ahead, though it still struggles with the two problems: efficiency and high production costs. In collaboration with Satoshi Uchida at the University of Tokyo, Michael Grätzel and his research group at the Swiss Federal Institute of Technology in Lausanne have now developed new sensitizers that should help an inexpensive type of solar cell to be more efficient. As they report in the journal Angewandte Chemie, the sensitizers are based on the dye indoline.
Some years ago, Grätzel developed photoelectrochemical solar cells that are inexpensive, easy to produce, and able to withstand long exposure to light and heat.
These "Grätzel cells" contain a mesoscopic layer of titanium oxide (TiO2) particles coated with a sensitizing dye. Upon irradiation with light, electrons are injected from the dye adsorbed on the TiO2, which are then transferred to the conducting band of the TiO2 and collected at the back contact, and carried away by an external circuit. In order for the cell to work, the electrons that are injected into the TiO2 must not recombine with the oxidized dye.
To prevent this, the cell contains an electrolyte solution with negatively charged iodide and triiodide ions as a redox couple dissolved in a solvent, which immediately reduce the holes created in the dye.
The main disadvantage of using volatile organic solvent in the electrolyte is the need for encapsulation of the electrolyte. Ionic liquids are an alternative to the use of these volatile solvent. These salts exist as liquids at low temperatures and do not evaporate. However, the high viscosity of these electrolytes is detrimental to the mass transport and consequently a problem for obtaining high efficiency.
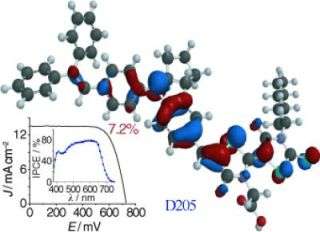
Grätzel and his team compensated for this loss of efficiency by optimizing the sensitizer. In place of the usual ruthenium dyes, they used tailor-made organic dyes based on indoline, which have a higher molar extinction coefficient. This allows the TiO2 films to be thinner, in turn reducing the electron path length. The combination thus attained an energy conversion yield of 7.2 %. This is a record for this type of cell (organic dye, ionic liquid, titanium oxide).
In this case the efficiency of the dye as a sensitizer is not only dependent on its chromophore, but also on its interfacial properties. So using a dye with an additional hydrocarbon chain has improved the performance by retarding the back electron reaction.
Citation: Michael Grätzel, Organic Dye-Sensitized Ionic Liquid Based Solar Cells: Remarkable Enhancement in Performance through Molecular Design of Indoline Sensitizers, Angewandte Chemie International Edition 2008, 47, No. 10, 1923–1927, doi: 10.1002/anie.200705225
Source: Angewandte Chemie


















