June 20, 2007 feature
Carbon nanotube injectors probe living cells without damage
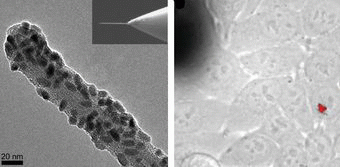
In order to investigate the processes that go on inside a single human cell—or even specific subcellular compartments—researchers need a device that is small and controlled enough to pass through the delicate cell membrane. Carbon nanotubes (CNTs), with their needle-like geometry, high elasticity and strength, have recently shown that they’re up to the task.
Scientists Xing Chen, Andrax Kis, Alex Zettl, and Carolyn Bertozzi from the University of California at Berkeley and the Lawrence Berkeley National Laboratory have recently found that a CNT-based “nanoinjector” is also the first to penetrate a cell with no membrane damage, even after hour-long, repeated use. Previous bulkier methods consistently damaged the membrane after just a few seconds of penetration.
“This is the first cell injector with temporal and spatial control on the nanometer scale that can deliver a discrete number of molecules and doesn't damage the cell membrane,” Chen told PhysOrg.com. “The less perturbation [to a cell], the better for the systems studied. In the case of cellular delivery/injection, if too much damage is made to the cell, the cell dies.”
The scientists predict that this advantage of the nanoinjector will play a central role in experimental cell biology with its ability to overcome the plasma membrane barrier. The nanoinjector also offers the opportunity for injecting cargo such as DNA, RNA, polymers, bacteria and other particles.
The key to the CNT’s success is simply its small size. With a diameter of 1 nanometer, about the same as a single protein, a CNT needle pierces a hole so small that it is quickly healed by lipid diffusion (the passive movement of particles to reach a state of equilibrium).
The setup consists of a multiwalled CNT attached to an atomic force microscope (AFM) tip. The AFM controls the needle’s displacement with nanometer resolution, as well as applies and monitors forces on the cell membrane.
In the scientists’ experiment, the CNT’s cargo consisted of quantum dots, which are fluorescent nanoparticles that provide high visibility to enable single particle tracking inside a cell. For loading and releasing this cargo, the scientists used a disulfide-based compound. In the oxidizing environment outside the cell, the disulfide is stable and links to the quantum dots; in the reducing environment inside the cell, the disulfide bonds are cleaved, releasing the quantum dots.
The scientists demonstrated this nanoinjection in cultured HeLa cells, which are human cervical epithelial cancer cells. After about 15-30 minutes of the nanoneedle positioned inside a targeted cell, the scientists observed 50-100 nanometer clusters of quantum dots using a fluorescence microscope.
Using single particle tracking, the scientists could also directly characterize the motions of the quantum dots inside the cell, which agreed with previous measurements. The long duration and repeated injection in living cells will hopefully make this biocompatible nanoinjector useful for scientists’ future investigations and delivery into single cells.
“Gene delivery is definitely one of the potential applications,” Chen said. “In addition, this new technique opens the door for studying a variety of intracellular processes.”
Citation: Chen, Xing, Kis, Andras, Zettl, A., and Bertozzi, Carolyn R. “A cell nanoinjector based on carbon nanotubes.” Proceedings of the National Academy of Sciences. 8218-8222, May 15, 2007, vol. 104, no. 20.
Copyright 2007 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.




















