How Titan's haze help us understand life's origins

Where did life on Earth come from? There are several theories as to what might have happened. Maybe comets came bearing organic material, or life was transported from another planet such as Mars, or something happened in the chemistry of our planet that made life possible.
Luckily for researchers, there is a possible laboratory in our solar system to help us better understand the conditions on Earth before life arose—a situation sometimes referred to as a "prebiotic" environment. That location is Titan, the largest moon of Saturn.
The moon has fascinated researchers for decades, particularly after NASA's Voyager 1 and Voyager 2 spacecraft flew by Saturn in the 1980s. The missions revealed a moon completely socked in with haze, which is a different experience to those used to gazing at Earth's airless, cratered moon.
A closer look came in 2004, when the Cassini-Huygens mission arrived to study the system. Since then, the spacecraft has done hundreds of flybys of Titan and peered at its surface by penetrating the clouds with radar. The European Space Agency's Huygens lander also made a soft landing on the moon in 2005.
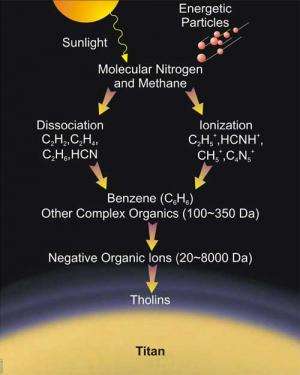
One of the big research questions is the composition of the haze. A new study is trying to recreate substances in the atmosphere called tholins, organic aerosols which are produced when radiation bakes the nitrogen and methane-rich atmosphere. In some cases, organics are considered precursors to life.
"The study of organic chemistry on Titan's surface would extend our understanding of the diversity of prebiotic chemistry, and perhaps life's origin on Earth," said Dr. Chao He, a chemist at the University of Houston (now moved to Johns Hopkins University) who led the study.
The results were publishedas "Solubility and stability investigation of Titan aerosol analogs: New insight from NMR analysis" in the journal Icarus.
Dissolving tholins
According to He, the study of Titan's tholins help scientists understand the basic properties of organic materials on Titan. Questions to consider include how they are structured, whether the aerosols can be dissolved in liquid in Titan's surface or atmosphere, and how stable the organics could be. Titan's tholins are thought to contain chemical precursors of life, and studying the molecule's structure helps scientists better understand whether life's possible precursors have formed on Titan. If they have formed, the solubility study helps to hint where to find them on Titan, and the stability study suggests the most capable detection methods.
The tholins were created by making a mix of methane (5 percent) and nitrogen (95 percent) in a reaction chamber at room temperature. The mixture was exposed to an electrical discharge for 72 hours, which then created a muddy substance—the tholin—on the walls of the vessel. The substances produced had a similar optical appearance to what Cassini observed in Titan's atmosphere.
Researchers then investigated how well the tholins would dissolve in a solvent. Several solvents were investigated, including polar solvents (methanol, water, dimethyl sulfoxide and acetonitrile) and non-polar solvents (pentane, benzene and cyclohexane). Polar solvents usually have different electrical charges between atoms (such as positive-charged oxygen and negative-charged hydrogen, in water) while non-polar solvents have similar electrical charges between atoms. Generally, polar solvents dissolve polar compounds best and non-polar solvents dissolve non-polar compounds best.
The researchers found that the tholins preferentially dissolves in polar solvents, suggesting little or none of the substance would be dissolved in the lakes or oceans on Titan, which are consist of non-polar ethane/methane. Thus, the tholins should be on the surface of the land or at the bottom of the lakes and oceans, He noted.
"The tholin preferentially dissolves in polar solvents, alsosuggesting the tholins are composed of a large percentage of polar species," Headded.
Picking future landing sites
The Huygens probe only survived on the surface of Titan for a few hours, but there are proposals out there to do extended missions. One example is a NASA Innovative Advanced Concepts proposal to send a submarine to explore Titan's lakes. The proposal is at the first stage of investigation and would be decades away to launch, if funding is approved.
If researchers were looking for tholins with a surface or underwater craft on Titan, He's study could help narrow down the location. Tholins break down in hot temperatures, but this is not a problem for Titan's surface, which sees an average surface temperature of -179 degrees Celsius (-290 degrees Fahrenheit). Future landing missions, however, might have to contend with avoiding heating the tholins to look at their structure, and instead should focus on nondestructive instruments and methods to accomplish this, He said. Possible methods of detecting organics could be liquid chromatography-mass spectrometry (LC-MS) and nuclear magnetic resonance spectroscopy (NMR). Both methods can provide detailed structural information of organic mixtures nondestructively.
Greater search for life's origins
In the midst of this analysis, He's team developed a new method to study the solubility of tholins. They found several nitrogenated organic molecules in Titan tholins.
"Some of them are very important to the prebiotic chemistry and the origin of life," He said.

While trying to understand what is happening on Titan, He is also interested in learning about the rest of the Solar System.
"My research focuses on the astrobiology on potential environments and objects," He said. "Titan is an important one. This study helps to understand the basic properties of organics on Titan. It also provides the basis for the development of in situ analysis of methods and instruments for a Titan mission and other outer planet exploration."
He plans to continue his study of organic chemistry on Titan, and then extend that understanding to other potentially interesting environments for life in the Solar System, such as Mars, Jupiter's icy moon Europa or Saturn's moon Enceladus, which has been recorded spouting water-rich plumes into the atmosphere.
Scientists' understanding of Titan is constantly changing as the Cassini-Huygens mission beams back data from the distant moon. For example, in 2007 scientists discovered that the tholins form at much higher altitudes than previously believed, at greater than 1,000 kilometers (621 miles) as opposed to a few hundred kilometers above the ground.

The results also revealed an unexpected high number of ions (negatively charged atoms) among the clouds of the moon, as well as detecting benzene, an element that is required to put together the tholins.
"The negative ions were a complete surprise," stated David Young, a scientist at the Southwest Research Institute in Texas, who led the Cassini Plasma Spectrometer (CAPS) investigation. "This suggests they may play an unexpected role in making tholins from carbon-nitrogen precursors."
A more recent finding revealed that Titan's atmosphere is likely older than that of Saturn. This suggests that the moon did not arise from the ringed gas giant, but instead was created separately in the gas and dust floating around the young Solar System while the Sun and planets were being formed.
Provided by Astrobio.net
This story is republished courtesy of NASA's Astrobiology Magazine. Explore the Earth and beyond at www.astrobio.net .


















