Designing new ways to probe aerosols for more accurate climate models
Dust storms, volcanic eruptions and hurricanes are tangible events that send particles of sand, soot and salt into the atmosphere. These particles can alter the climate cycles that heat and cool the Earth. Less obvious sources of aerosols affect these cycles, too; the sea salt sprayed off breaking waves or volatile organic compounds emitted from trees. But nearly untraceable chemical transformations also lead to the birth of new atmospheric particles. Accounting for these newborn particles and their role in cloud formation is key to improving climate models. Scientists recently gathered at EMSL to discuss ways to more accurately chronicle the clandestine lives of these aerosols in the lab and the field.
The precise processes that govern how new particles are created out of vapor, then grow and nucleate clouds – enticing other organic molecules into chemical reactions – largely remains a mystery. Increasingly sophisticated instruments provide scientists with new details about these transformations; they also point out big gaps between theoretical models and field findings.
"We discovered nucleation is an important source of atmospheric particles more than 20 years ago. And ever since we've been trying to figure out the chemical and physical processes that lead to their creation," says Peter McMurry, a professor at the University of Minnesota. "The goal is to achieve closure between the rates observed in the atmosphere and those predicted by models. This will lead to more credible climate models. Right now we can't do that."
In the Beginning
Part of the problem is catching the particles in the act of doing whatever it is they do. Researchers spend weeks in the field stalking new particle formation events. With little lead time the particles are born. Within a few hours they grow to sizes large enough to serve as cloud condensation nuclei, sending a brief bloom of data across suites of specialized sampling instruments. "It's a challenge," says McMurry. "Some of the species that lead to particle formation are found in almost unimaginably low concentrations. Sometimes it's like trying to pick out one person in a thousand or a million Earths."
From Field to Lab: An Inside Job
For the last 15 years Alexander Laskin, an atmospheric aerosol chemist at EMSL, and collaborators have been using the unique analytical chemistry capabilities of EMSL on particle samples collected from test facilities, laboratory studies and far-flung field sites. A variety of off-line chemical imaging techniques have been advanced at EMSL – including mass spectrometry, spectroscopy, microscopy and microanalysis methods – to probe the composition and properties of these particles. But there's no single technique in existence that gives us all the information we need to know. says Laskin"
The ability to run multi-modal analyses on the same sample is a particular strength at EMSL. But even interrogating particles with multiple techniques can't answer some of the pressing questions. So EMSL researchers are devising new strategies in collaboration with scientists across the country.
Laskin and his colleagues want to understand the physical and chemical properties of aerosols that nucleate ice clouds. Although ice nucleation has been studied for more than two decades, much about the process remains unknown.
"In the traditional view of ice nucleation events, there are very few particles in the atmosphere with the right properties to become ice nuclei," says Laskin. "But now there's growing evidence that's not always case," he says. "Particles with mediocre ice nucleation propensity – but present at high number concentrations – can play an equivalently important role. So the big question is how to delineate the two scenarios: Under which conditions would different nucleation particles prevail?"
It would be easier to figure out if they could visualize nucleation events, says Bingbing Wang, a postdoctoral researcher at EMSL. To see which individual particles nucleate ice first, he started a project coupling ice nucleation experiments with scanning electron microscopy, complemented with particle spectro-microscopy. Then he can probe the composition of the instigating particles. "From that knowledge we can achieve better understanding of heterogeneous ice nucleation. Then with further data analysis we can provide better parameterizations for cloud and climate models," says B. Wang.
The study involves a group of Pacific Northwest National Laboratory experts in collaboration with Daniel Knopf of Stony Brook University and Mary Gilles at Lawrence Berkeley National Laboratory.
An Outside Approach
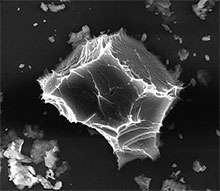
Another way to analyze these perplexing particles is to look at them one by one. To accomplish this, transmission electron microscopy, or TEM, is the technique of choice for Peter Buseck, a professor at Arizona State University who attended the recent user meeting at EMSL. "We want to know the intimate details about these aerosol particles," he says. "TEM has been used on a larger scale, but now the technique is needed on a nanometer scale because there's a whole range of particle sizes in the atmosphere."
Although Buseck already has the TEM capabilities he needs, EMSL could devise novel ways to combine electron microscopy with instruments such as the helium ion microscope in EMSL's Quiet Wing. No one's doing that now with aerosols, says Buseck.

Probing the Surface
It's not just the particles that need to be accounted for.
"An intrinsic scientific problem is how reactions occur and the particle surface is key for those. From mass spectrometry you can find out what kind of molecules are in aerosols, but you don't know how they are arranged on the particle surface," says Hongfei Wang, a chief scientist at EMSL.
To get at the structure of the surface, in 2011 H. Wang and his colleagues developed a unique high-resolution sum frequency generation, or HR-SFG, spectrometer to discern surface particle reactions with a resolution 10 times better than any other instruments out there. That kind of resolution was critical for Sergey Nizkorodov who studies aerosol photochemistry in his laboratory at the University of California at Irvine and then sends samples to H. Wang for high-resolution studies at the surface.
"We want to be able to detect minor species that might be important for controlling reactions," says H. Wang. "We've improved resolution, but the sensitivity of the signal needs to be improved for field measurements. It's possible – but will take at least several years. It will be a real milestone to do this."
Another pioneer in applying SFG spectroscopy to aerosol particles is Franz Geiger of Northwestern University. After meeting H. Wang at an American Chemical Society conference two years ago, he started using the high-resolution instrument to study alpha-pinene, one of the main components of secondary organic aerosol particles. EMSL's HR-SFG gave Geiger a significant improvement in his analytics, getting resolution from standard values of about 10 wave numbers down to 0.6 wave numbers.
To bridge the gap between particle analysis in the lab and the field, Geiger is working with Northwestern colleague Regan Thomson, Victor Batista of Yale University and Scot Martin at Harvard University on an EMSL user project and with matching funds from Northwestern (and he hopes, the National Science Foundation).
Geiger is focused on the organic emissions of trees, namely pinene, that react with ozone at the ground level. The resulting cascades of reactions create aerosols that can interact with water vapor and ultimately make clouds. The questions he wants to answer are: Will future, possibly warmer, climates lead to faster emission rates of pinene over the Northern hemisphere? And if more organic particles form, potentially leading to more clouds, would this cause a negative feedback mechanism to cool our planet?
The answers to how these particles form will be found on their surfaces, thinks Geiger. But it's hard to analyze what's never been identified before. So the collaborators are creating synthetic compounds they surmise could be created when pinene reacts with ozone and are also surface reactive. By analyzing the chemical signatures of these lab-made components they'll build a reference library to help identify the unknown surface components they discover on these particles.
"There's no silver bullet," says Geiger. "But more current tools will give us a more complete understanding."

Helping Solve Problems
Aerosol research may also get a boost with instruments that haven't traditionally been used for studies in this field. In the past, the tiny sample sizes and filter collection methods for aerosol particles have hampered efforts to use analytic techniques such as nuclear magnetic resonance, or NMR, spectroscopy. But at the recent EMSL user meeting, NMR lead scientist Karl Mueller told researchers they're building smaller and better tools to aid aerosol research.
"NMR is phenomenal for determining chemical structure," says Nancy Washton, the capability lead for EMSL's NMR group. "But it's considered an insensitive technique because it requires such a large sample size – I want a vial full of stuff, about 20 milligrams."
Now Mueller and Washton are working with a team of scientists to make very small volume NMR detectors. "We're combining our abilities as 'spin jocks' with miniaturization and microfabrication capabilities to work with picoliter amounts more suitable for aerosol studies. Success here would be a real breakthrough," says Mueller.
Washton anticipates they'll be able to run aerosol samples within the year. The possibilities have already enticed collaborators to send samples and she thinks NMR will be very useful for scientists who are synthetically producing atmospheric molecules.
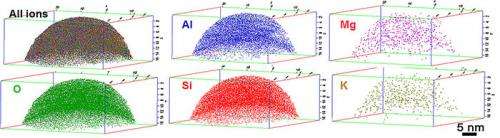
Another technique that might prove useful is atom probe tomography, or APT. EMSL Scientist Arun Devaraj thinks APT could provide a way to visualize the three-dimensional element distribution within a single aerosol particle at sub-nanometer spatial resolution.
"Even if a potential user doesn't quite know what capabilities they need, the staff at EMSL can discuss their needs and provide guidance," says Alex Guenther, the lead scientist for atmospheric aerosol science at EMSL. The staff will make extraordinary efforts to understand what scientists need for their research and how the laboratory's capabilities can be applied to answer their scientific questions, he says.

Although new tools are nice, Nicole Riemer, a computer modeler at Illinois University, thinks researchers may squeeze more information out of the tools already on hand. "As modelers, we are using different tools from the researchers in the lab who measure these things," says Riemer. "But we're trying to solve the same problems."
The aerosols are really, really small. So it's difficult to develop equations that reflect the complexity of those minuscule – but critical – interactions without bogging down the ability to scale up to global proportions. Riemer believes careful, repeated experiments under varied conditions, using the equipment already in EMSL laboratories, can provide key details needed to derive more accurate aerosol predictions. For example, our models are based on particles with spherical shapes, says Riemer. Yet Laskin's work shows particle morphology changes. "Is that important?" she says. "If it is, we need to know how it changes over time so we can incorporate it into our equations."
Gathering Better Field Samples
But even the best available laboratory instruments don't really capture the conditions in the atmosphere. So, Guenther invited scientists to brainstorm better ways to get samples from the field. Among the more immediately feasible possibilities discussed were ways to build a better Orbitrap.
"I was excited to hear discussions about making a smaller high-resolution mass spectrometer to carry around in a truck or plane," says Nizkorodov. "If there's a way to get those measurements in real time, that would be a big step forward."
With that kind of innovation, Franz Geiger may one day realize a decades-old research dream: to analyze aerosol particle surfaces without plucking them out of the air. "People once said we couldn't study aerosols from ships or planes and now we do that all the time," he says. "It's not impossible."
Provided by Environmental Molecular Sciences Laboratory