February 1, 2013 report
Nanoscale MRI being developed

(Phys.org)—Two independent groups of scientists in the U.S. and Germany have reduced magnetic resonance imaging (MRI) down to the nanoscale, which may enable them in the future to non-destructively detect and image small molecules such as proteins at room temperature and pressure. Previously, nanoscale imaging was only possible at extremely low temperatures and pressures.
MRI works through detecting weak electromagnetic fields produced by the nuclei of atoms such as hydrogen within the molecules being studied, and the collective resonance of these fields. It is able to image structures without destroying them, which makes it useful for scanning bodies, but its relatively low sensitivity has up to now restricted its use on the small scale to chemicals with volumes measured in micrometers at best.
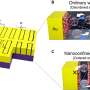
Two papers published in the journal Science describe the research done by the two separate groups, which both used dark spots, or nitrogen-vacancy (NV) defects, on the surface of diamonds. Diamond is inert magnetically because it consists entirely of carbon atoms bonded covalently, and there are no free electrons. However, there can be imperfections such as NVs, in which a single carbon is replaced by a nitrogen atom, adjacent to a vacancy in the lattice where a carbon atom is missing. The NVs have a free electron, which gives it unique magnetic properties, and it is these properties the two research teams exploited.
The first team, led by Daniel Rugar and John Mamim of the Almaden Research Center in San Jose, California, used diamond dark spots to detect weak magnetic fields in materials near the diamond surface. Rugar's group synthesized extremely pure diamond with NV centers close to the surface and overlaid it with a polymer 60 nanometers thick. They then applied an oscillating magnetic field. Dr. Rugar explained that when you shine green light on the dark spots they fluoresce in red, and the brightness depends on the NV center's magnetic state. External magnetic fields in the vicinity can affect the spin of the NV center electron, which in turn affects the brightness of the fluorescent red.
The second team, led by Friedemann Reinhard of the University of Stuttgart, also used nitrogen-vacancy defects on extremely pure samples of synthesized diamond, but they used them to record the NMR spectra of a range of chemicals placed on the surface of the diamond. Dr. Reinhard said their method was more passive than the methods used by Rugar's team, but this makes it a little easier to implement.
The research is important because it is difficult to determine protein structures conventionally, which involves expressing and purifying the proteins and then crystallizing them. Being able to take an MRI image would simplify the process and enable the structures of all proteins to be worked out. At the moment the research of both teams is at a "proof of principle" level, according to Rugar's team, and more research is needed before the techniques can be used to image molecules.
More information: 1. Nanoscale Nuclear Magnetic Resonance with a Nitrogen-Vacancy Spin Sensor, Science 1 February 2013: Vol. 339 no. 6119 pp. 557-560 DOI: 10.1126/science.1231540
ABSTRACT
Extension of nuclear magnetic resonance (NMR) to nanoscale samples has been a longstanding challenge because of the insensitivity of conventional detection methods. We demonstrated the use of an individual, near-surface nitrogen-vacancy (NV) center in diamond as a sensor to detect proton NMR in an organic sample located external to the diamond. Using a combination of electron spin echoes and proton spin manipulation, we showed that the NV center senses the nanotesla field fluctuations from the protons, enabling both time-domain and spectroscopic NMR measurements on the nanometer scale.
2. Nuclear Magnetic Resonance Spectroscopy on a (5-Nanometer)3 Sample Volume, Science 1 February 2013: Vol. 339 no. 6119 pp. 561-563 DOI: 10.1126/science.1231675
ABSTRACT
Application of nuclear magnetic resonance (NMR) spectroscopy to nanoscale samples has remained an elusive goal, achieved only with great experimental effort at subkelvin temperatures. We demonstrated detection of NMR signals from a (5-nanometer)3 voxel of various fluid and solid organic samples under ambient conditions. We used an atomic-size magnetic field sensor, a single nitrogen-vacancy defect center, embedded ~7 nanometers under the surface of a bulk diamond to record NMR spectra of various samples placed on the diamond surface. Its detection volume consisted of only 104 nuclear spins with a net magnetization of only 102 statistically polarized spins.
Journal information: Science
© 2013 Phys.org