Heavy Pyridine Crystallizes Differently
(PhysOrg.com) -- The nuclei of ordinary hydrogen atoms contain only a single proton. If a neutron is added, the hydrogen becomes deuterium. In principle, molecules that contain deuterium in place of hydrogen atoms are chemically identical. However, there can be significant differences. Thus “heavy water”, water with molecules that contain deuterium in place of hydrogen, is toxic because it disrupts highly sensitive biochemical processes in the body and leads to metabolic failure.
As researchers report in the journal Angewandte Chemie, when the hydrogen atoms of pyridine are replaced with deuterium, it adopts a crystalline form that can only be achieved under high pressure with “normal” pyridine. Perhaps the minimal differences responsible for this type of effect can be implemented to improve the spectrum of properties available to pharmaceutical agents.
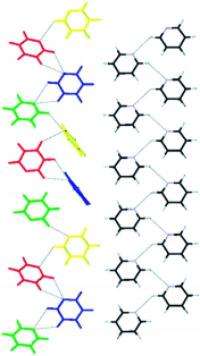
Pyridine is a six-membered ring with five carbon atoms and one nitrogen atom. The carbon atoms are each attached to one hydrogen atom. These can be replaced with deuterium. Researchers led by Roland Boese at the University of Duisburg-Essen have discovered that deuterated pyridine crystallizes at about -85 °C with a different crystal structure than that usually adopted by pyridine. In parallel, British researchers working with Simon Parsons determined that non-deuterated pyridine also adopts this structure under high pressure, because it occupies a smaller volume than pyridine’s usual structure.
The replacement of hydrogen by deuterium clearly changes the strength of interactions between individual groups of atoms in neighboring molecules, making other arrangements more energetically favorable. Such interactions between groups of atoms also play an important role in pharmaceuticals, such as when a drug is meant to fit into the binding cavity of an enzyme. Subtle changes can result in significant changes in a drug’s activity.
This is why Boese and his team are interested in deuterated pyridine: pyridine is an important starting material for pharmaceuticals, and its basic framework is found in many medications. Boese thinks it likely that deuteration will allow for the development of drug variants that are more specific or have fewer side effects than their conventional precursors.
Paper: Roland Boese, Isotopic Polymorphism in Pyridine, Angewandte Chemie International Edition 2009, 48, No. 4, 755-757, doi: 10.1002/anie.200803589
Provided by Wiley