Researcher working on destruction of chemical weapons
America's war on terror includes fighting the dark side of deadly chemical agents, and Texas A&M University chemist Dr. Frank Raushel is helping with the fight by developing an enzyme that might neutralize one such chemical agent, the organophosphates.
Synthetic organophosphates started their journey as insecticides in the 1930s but soon made their way into the dangerous alleys of chemical warfare during World War II. Raushel, who has been working on detoxification of organophosphates for nearly 10 years, says that the toxic properties of the organophosphate nerve agents are a serious threat to the health and well being of civilized societies.
Although outlawed by several international treaties, some countries have used chemical agents during times of war. In 1988, the Iraqi Kurdish village of Halabja was exposed to multiple chemical agents, killing about 5,000 of the town's 50,000 residents. After the incident, traces of mustard gas and the organophosphates sarin, tabun and VX were discovered.
Raushel, the Davidson Professor of Science at Texas A&M, has been awarded a four-year grant of $1.2 million by the National Institutes of Health to carry out his work on organophosphates.
Organophosphates are neurotoxins — they attack the nervous system by blocking the function of the enzyme acetylcholine esterase, which carries nerve signals. When organs such as the lungs do not receive appropriate nerve signals, control is lost over respiratory muscles, and it usually results in death by asphyxiation. Today, some classes of organophosphates are used as insecticides while others have been categorized as chemical weapons.
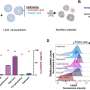
Researchers have discovered a bacterial enzyme, phosphotriesterase, which can recognize and destroy the toxicity of a broad spectrum of organophosphate nerve agents. What Raushel aims to do is design and characterize bacterial phosphotriesterases that are better at detecting, destroying and detoxifying those organophosphates that pose the most serious threats to human health.
Source: Texas A&M University