New Nanoparticles for Targeting Tumors
As a wide variety of nanoparticles continue to demonstrate their ability to improve the delivery of imaging agents and drugs to tumors, nanoparticle researchers have turned their attention to the challenge of systematically determining how a given nanoparticle’s physical and chemical characteristics affect its ability to target tumors. Such data could provide drug developers with guidelines to help them select the most effective type of nanoparticle for a given therapeutic or imaging application.
In a paper published in the PNAS, a team of investigators at the MIT-Harvard Center for Cancer Nanotechnology Excellence, led by Omid Farokhzad, Ph.D., at the Harvard Medical School, and Robert Langer, Ph.D., at MIT, describe one such approach to systematizing nanoparticle development.
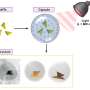
In their research, the investigators created used two self-assembling polymer families to create series of tumor-targeted nanoparticles that varied slightly from one another in terms of their physical characteristics and their biopharmaceutical properties.
By changing the exact composition of each of the two polymers, as well as the ratio of the two polymers, the investigators found that they could fine-tune both the size and drug-releasing properties of the nanoparticles, which were targeted to the prostate-specific membrane antigen found on the surface of prostate cancer cells. The researchers also were able to vary the amount of targeting agent on the nanoparticle surface, as well as the “stealth” characteristics of the nanoparticle, that is, the ability to evade the immune system. By studying the effect of each change on nanoparticle uptake by prostate cancer cells growing in tissue culture, the investigators were able to identify the specific formulation that optimized tumor uptake in vivo.
Investigators at the University of California, Berkeley, and the University of California, San Francisco, achieved similar results with a different class of polymer nanoparticles known as dendrimers. Jean Fréchet, Ph.D., at UC-Berkeley, and Francis Szoka, at UCSF and a member of the Carolina Center of Cancer Nanotechnology Excellence, led the team of collaborators that created libraries of dendrimers containing a variety of functional groups on their surfaces. These functional groups enable the investigators to attach both PEG and any number of targeting, imaging, and therapeutic agents to the dendrimer surface in a systematic manner.
Experiments using radiolabeled dendrimers demonstrated that these nanoparticle were able to circulate in blood for long periods of time. Subsequent experiments using a dendrimer linked to the antitumor agent doxorubicin showed that drug-loaded carrier accumulated in tumors but far less in healthy tissue compared with liposomal doxorubicin, the first nanoparticle-based drug approved to treat cancer.
The work from Drs. Farokhzad and Langer, which was supported in part by the NCI’s Alliance for Nanotechnology in Cancer, is detailed in the paper “Precise engineering of targeted nanoparticles by using self-assembled biointegrated block copolymers.” An abstract of this paper is available through PubMed.
The work from Drs. Fréchet and Szoka is detailed in the paper “PEGylated dendrimers with core functionality for biological applications.” An abstract of this paper is available through PubMed.
Source: National Cancer Institute