Mixed conductor ceria proven as excellent catalyst for fuel cell anodes
Researchers at the California Institute of Technology have discovered that ceria (or cerium dioxide) is an excellent catalyst for fuel cell anodes. This discovery was made through careful electrochemical characterization using a.c. impedance spectroscopy, an important tool for studying the electrical transport behavior of ceramics. The study is published in the Journal of the American Ceramic Society.
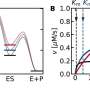
By applying an a.c. electrical voltage to the ceria (a white crystalline powder used in the manufacture of ceramics) and measuring the a.c. electrical current, researchers demonstrated they could precisely quantify the amount of the electrical current carried by the electrons. This information is critical for any application researchers might have in mind for materials which ideally conduct only ions, and, in particular, as the electrolyte in fuel cells.
"This will likely change the way that people perform and interpret the results of a.c. impedance spectroscopy measurements," states Professor Sossina Haile, one of the study researchers. "It turns out that there is more information to be had than people previously understood."
It was further discovered that for the reaction of H2 gas with oxygen ions from the ceramic to form H2O, ceria behaves as its own catalyst. This is significant because this hydrogen electro-oxidation reaction is exactly what takes place in a fuel cell anode. Getting fuel cells to perform efficiently requires getting such electrochemical reactions to occur rapidly.
"There had long been suspicion that ceria was somehow a good thing in the [fuel cell] anodes," adds Professor Haile. "This now clearly explains why and how, and puts the observations beyond debate."
Source: Blackwell Publishing Ltd.