New Way to Synthesize Quantum Dots for ' Biological Tags'
A University at Buffalo research team has invented a new way to synthesize quantum dots -- luminescent nanocrystals made from semiconductor material.
Sometimes called artificial atoms, quantum dots have the potential to be used to build exciting new devices for biological and environmental sensing, quantum computing, lasers and telecommunications, among other applications.
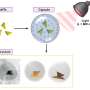
The new technique developed by a team led by T.J. Mountziaris, Ph.D., professor of chemical and biological engineering in the UB School of Engineering and Applied Sciences, enables precise control of particle size by using a microemulsion template formed by "self-assembly." The process involves the direct mixing of a nonpolar substance (heptane), a polar substance (formamide) and an amphiphilic substance or surfactant (a block copolymer) to form a uniform dispersion of heptane droplets in formamide, stabilized by the surfactant.
A patent is pending on the technique, which was described in a recent issue of the journal Langmuir. Mountziaris' co-researchers are Paschalis Alexandridis, Ph.D., UB professor of chemical and biological engineering; Athos Petrou, Ph.D., professor of physics in the UB College of Arts and Sciences; Georgios Karanikolos, a graduate student in the UB Department of Chemical and Biological Engineering; and Grigorios Itskos, a graduate student in the UB Department of Physics.
Using the technique, the UB researchers demonstrated the controlled synthesis of zinc selenide (ZnSe) quantum dots that exhibit size-dependent luminescence. When excited by ultraviolet light, quantum dots emit a particular fluorescent color and brightness, depending on the dot's size. The problem for scientists has been devising simple techniques to control the size of quantum dots, which would give them the ability to control a quantum dot's color properties. Such control is a critical factor in the quantum dot's functionality.
The ZnSe quantum dots have potential for use in clinical and therapeutic diagnostics and for DNA analysis. The dots may be used, for example, as biological tags, attaching themselves to diseased cells, tumors or particular genes, alerting scientists to their presence in the body or in biological samples.
"The luminescent properties of quantum dots make them ideal for such applications," Mountziaris explains.
The technique developed by Mountziaris and co-researchers gives them the ability to precisely control the size (and luminescence wavelength) of the ZnSe dots in one step. The researchers were able grow ZnSe dots inside "nanoreactors" formed by the heptane nanodroplets of the emulsion. By reacting hydrogen selenide gas with diethyl-zinc (DEZn) dissolved in the heptane, a single quantum dot is grown in each nanoreactor, allowing precise control of particle size by simply controlling the initial concentration of DEZn in the heptane. Small clusters of ZnSe nucleate in each heptane nanodroplet and fuse into one particle by a process called coalescence. The researchers run the process at room temperature, but still obtain crystalline particles.
"Since we run the process at room temperature, we were expecting amorphous particles or crystalline particles with many defects. To our surprise, we obtained almost perfect crystals," Mountziaris says. "We believe that the localized energy release during cluster-cluster coalescence is the key to forming single crystalline particles.
"The energy released anneals the particles and leads to perfect crystals," he adds.
ZnSe quantum dots created by this technique have maintained their luminescent properties for more than a year.
To make quantum dots useful for practical applications, functional molecules must be attached to their surface after they are synthesized, Mountziaris explains.
"Researchers are creating biological tags of certain colors based on quantum dots by decorating their surface with functional molecules that selectively attach to a specific biological molecule," he says. "This gives the molecules something like a tail light, and you could follow them in the body by exciting their luminescence with ultraviolet light."
Mountziaris' group is collaborating with UB bioengineers to use quantum dots in DNA analysis.
"The challenge of quantum-dot technology has been how to make dots of a precise size, how to functionalize the surface and also scale up the process for commercial applications," Mountziaris says. "Our technique can be scaled up very easily because it is based on self-assembly and does not depend on mixing efficiency or process time to control the size of the dots. We have demonstrated 'dial-a-size' capability."
"One nanoreactor makes one quantum dot," he adds. "My colleague, Paschalis Alexandridis, and our student, George Karanikolos, have developed a very stable microemulsion that has very slow droplet-droplet interactions. This prevents agglomeration of the nanocrystals after they are formed, which can adversely affect their properties. It is also responsible for the remarkable stability of the quantum dot loaded emulsion."
Mountziaris and co-researchers are at work synthesizing additional compounds, such as cadmium selenide and lead selenide to cover a wide spectrum of luminescence wavelengths. They also are developing functional water-soluble caps for the quantum dots that would enable their use as biological tags, without diminishing the dots' luminescence.
Multicolor quantum dots could be used to create "optical bar codes" from a sequence of joined quantum dots possessing different luminescent properties, Mountziaris says. "This would be very useful in multiplexed experiments by assigning a different function to different groups of dots and tracking them as they attach to different biomolecules," he explains.
The University at Buffalo is a premier research-intensive public university, the largest and most comprehensive campus in the State University of New York. UB's more than 27,000 students pursue their academic interests through more than 300 undergraduate, graduate, and professional degree programs.
Source: University at Buffalo