Cell map of human gonads identifies the cells involved in sex determination
The first large-scale cellular map of gonadal developmental in both sexes has been created by researchers at the Wellcome Sanger Institute, as part of the Human Cell Atlas initiative to map all cell types in the human body.
The findings, published today in Nature, identified new cell types including those that express the "sex determination" gene, beginning the process that decides whether an individual will become phenotypically male or female. The map will be transformative in improving the culture of gametes in fertility treatments and understanding reproductive conditions, such as differences in sex development.
The gonads play a key role in human development. They determine biological sex before maturing into ovaries in females or testes in males, which produce the egg and sperm cells required for reproduction.
The early weeks of development are extremely dynamic, with cell types appearing and disappearing rapidly as their purpose is fulfilled. This makes it challenging to study the events that lead to sex determination and subsequent differentiation into testis- or ovary-specific cells. While most of our knowledge of gonadal development comes from mouse studies, it is uncertain how much of this knowledge can actually be translated to humans.
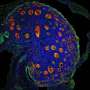
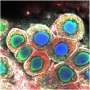
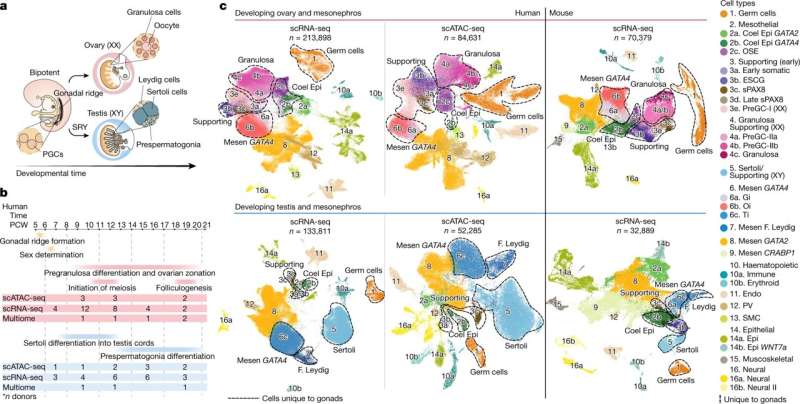
In this new study, researchers at the Wellcome Sanger Institute set out to create the first large-scale cellular map of gonadal developmental in both sexes, in order to characterize the route that cells take to become either a testis or an ovary during early life. They analyzed around a half-million cells from human gonadal tissue using single-cell sequencing and spatial transcriptomics, covering weeks six to 21 of pregnancy.
The team also generated a similar map in mice that they used to understand where human and mouse biology are the same or different. To date, this is the most detailed map of the developing gonads in terms of both time and space.
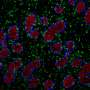
The combination of single-cell and spatial technologies enabled the researchers to identify not only which genes were expressed in individual cells, but also to understand the arrangement of the different cell types within the tissue and how cells talk to one another. This is key to decipher how the developing gametes interact with their neighboring cells as they mature inside the gonads to become sperm or egg cells.
The team identified patterns of gene expression that are unique to humans and are not shared with mice. Notably, the researchers pinned down the cell type that is the first to express the "sex determination" gene, which initiates the process to decide whether the undifferentiated gonad will become a testis or an ovary. Named Early Supporting Gonadal Cells (ESGCs), this cell type was found to peak around six weeks after conception. ESGCs are present in both humans and mice, yet their gene expression pattern is different in the two species.
"Gonadal development is a complex process and it is only at single-cell resolution that you start to see all of the cell types involved in sex differentiation and can narrow down the timeframe in which this process occurs. The characterization of Early Supporting Gonadal Cells and their role in kick-starting sex determination is an exciting discovery that will help researchers to better understand this crucial time in human development," says Dr. Luz Garcia-Alonso.
Another key finding was specific populations of macrophages, a type of immune cell, in the testes. One of these populations resembled macrophages found in the brain, while another resembled those found in bone.
"It was fascinating to find macrophage populations in the gonads that we're used to seeing in other organs, considering the very different purposes of the testes, the brain and bone. However, drawing parallels between the immune requirements of each organ can help us to understand the role of these macrophage populations wherever they occur in the body. In terms of evolution, if you have a biological function that could be useful elsewhere, why not use it," says Valentina Lorenzi.
One of the applications of these data will be to better understand differences in sex development (DSD). Although DSD has recently been in the headlines in relation to sport, it encompasses a wide array of situations where a person's chromosomal, gonadal or anatomical sex is atypical. The team's effort in extracting human-specific gene expression patterns of the cell types appearing during early gonadal development will prove instrumental to improve the management of DSD.
"This large-scale cellular map of gonadal development is a valuable addition to the Human Cell Atlas initiative that will enable research in this intriguing and complex area of human biology. I fully expect these data to further our understanding of differences in sex development, infertility and reproductive diseases," says Dr. Roser Vento-Tormo.
More information: Luz Garcia-Alonso et al, Single-cell roadmap of human gonadal development, Nature (2022). DOI: 10.1038/s41586-022-04918-4
Journal information: Nature
Provided by Wellcome Trust Sanger Institute