Field study reveals how ammonia isotope molecules diffuse in air

Scientists are working to find ways to monitor ammonia—an increasingly common species in the atmosphere closely linked to water eutrophication, soil acidity, and biodiversity loss. But measuring ammonia is not easy because ammonia can change from gas to particles quickly in the air.
Although there are accurate methods to determine ammonia in the lab with the aid of a pump, they are unsuitable for long-term measurements and for large-scale surveys in the field, due to the cost, particularly in terms of manpower, and the need for constant power supply.
To overcome these shortcomings, an alternative method was developed to collect ammonia from the atmosphere without the aid of a pump. The method is called passive sampling, in contrast to previous methods using a pump. It can provide a simple and cost-effective tool for monitoring ammonia without power requirement.
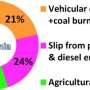
However, in a study published in Atmospheric Research on May 3, scientists tested three passive samples used in ammonia collection, and found that the ammonia concentrations were all subject to low bias (13.4-27.5%), compared with a standard reference method using a pump to draw air into an active sampler.
To address this issue, the researchers used the classic theory of Fick's law to revisit the effective flow rate of how much ammonia will diffuse onto an acid-coated filter housed in the protective cases of the passive samplers.
The theoretical flow was then temperature- and pressure- corrected with local observations during the sampling campaign in urban Beijing.
"We found the effective sampling flow rate of the passive samplers were all lower than the nominal value provided by the manufacturer," said Yuepeng Pan from Institute of Atmospheric Physics (IAP) of the Chinese Academy of Sciences, lead author of the study. "The inaccurate effective sampling rate in the passive collection samplers was suggested to be the result of incorrect mass transfer correction factors that were not well considered by the manufacturer."
While the underestimation of ammonia concentrations by passive samplers was previously reported, the ammonia isotope compositions (15N/14N) collected by passive samplers were arguably surprising lower than the active reference method, with a large difference of 15.4‰ in the field investigation of urban Beijing.
Prior to this study, scientists had little confidence to tell which data—collected by passive or active samplers—are more suitable in characterizing ammonia isotope composition. But now the low bias can be corrected by adding 15.4‰ to the value of the ammonia isotope compositions obtained from passive samplers in previous studies.
"The large off-set of 15.4‰ was due to a diffusion isotope effect between light ammonia isotope (14N) and heavy ammonia isotope (15N), driven by relative mass differences (one gram per mole)," said Pan. "I believe this study offers a firmer handle on characterizing ammonia sources and therefore enhances our ability of quantifying which emissions may affect increasing ammonia in future."
More information: Yuepeng Pan et al, Systematic low bias of passive samplers in characterizing nitrogen isotopic composition of atmospheric ammonia, Atmospheric Research (2020). DOI: 10.1016/j.atmosres.2020.105018
Provided by Chinese Academy of Sciences