Study shines light on spread of Candida auris
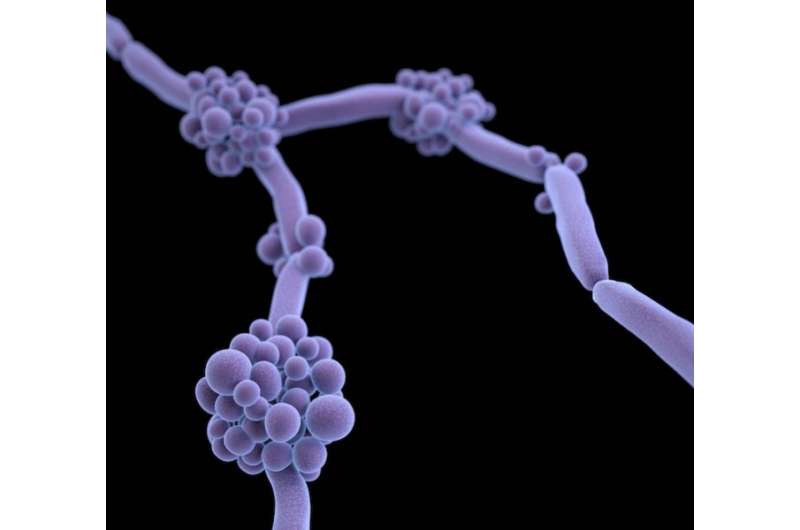
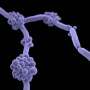
Candida auris is capable of forming high burden biofilms, which may help explain why this fungal pathogen is spreading in hospitals worldwide, according to a study published this week in mSphere, an open-access journal of the American Society for Microbiology. The research also establishes a new model to investigate the spread of this emerging fungal pathogen that causes invasive infections and is an emerging problem in hospitals worldwide.
"With these findings, we are able to have new tools to examine how Candida auris forms biofilms and spreads in this setting. Understanding more about this process could help us develop new strategies to prevent biofilm formation," said Jeniel Nett, MD, Ph.D., the study's principal investigator and assistant professor in the Departments of Medicine and Medical Microbiology & Immunology at the University of Wisconsin School of Medicine and Public Health. "Our tools can be used to determine how the biofilms are formed, which may lead to new strategies to prevent the transmission of or pharmaceutically target Candida auris."
Unlike many other Candida species, Candida auris spreads rapidly among patients and is efficient at colonizing skin. Because many of the patients infected with Candida auris have indwelling medical devices such as vascular catheters and endotracheal tubes, the researchers hypothesized that biofilms may be forming on these devices and on skin, facilitating transmission of Candida auris. "Candida species, similar to other fungal species and bacteria, can form communities that are adherent to a substrate, either on an artificial device or other surface," said Dr. Nett. "These biofilms communities are multilayered, and they often resist medical treatment, including antifungals, and host responses."
In the new study, Dr. Nett and colleagues developed a synthetic sweat media to represent skin conditions and using this model, they evaluated the growth of Candida auris and Candida albicans, one of the most common Candida species. They also examined the growth of these Candida species using a pig skin model to represent human skin conditions.
The researchers found that Candida auris exhibited a heightened capacity to both grow in the skin niche condition and to colonize pig skin and that Candida auris grew and colonized pig skin by forming multilayer biofilms. "Candida auris can grow approximately tenfold greater as a biofilm in this setting when compared to Candida albicans, one of the more common Candida species that does not seem to spread in hospital settings in this way," said Dr. Nett. "We think that this mode of growth is how Candida auris propagates in healthcare settings. Candida auris appears to colonize skin exceptionally well, growing particularly well in skin niche conditions."
The researchers said the formation of the biofilms is one of the reasons why Candida auris is difficult to clean in hospital settings. "These biofilms are likely on a lot of medical equipment used in healthcare settings," said Dr. Nett.
The researchers said their next steps are to further develop their model and examine Candida auris growth to determine some of the triggers for how Candida forms biofilms. "I was surprised that Candida auris formed such a condensed biofilm in this skin setting, as other species did not," said Dr. Nett. "We hypothesized that Candida auris may have a capacity to replicate in this environment, but we didn't expect it would be tenfold greater than the other Candida species."
Provided by American Society for Microbiology