Highly efficient cooling using a new nanoporous solid
Heat recovery (solar energy, heat pump, air conditioning, cooling) is a key research focus toward reducing power consumption and encouraging sustainable development. Even if water recovery and release using nanoporous materials is a reliable strategy to achieve this goal, developing new energy-efficient processes remains a challenge. Researchers from the Paris porous materials Institute (CNRS, ENS Paris, ESPCI Paris/ PSL University) and from the Charles Gerhardt Institute in Montpellier (Université Montpellier/CNRS/ ENSCM) have discovered a new hybrid porous material that is robust and synthetized through a "green chemistry" route. In Nature Energy, they report that this new material is much more efficient than any other water adsorbent, with a high storage capacity and a lower regeneration temperature.
The use of water sorption (i.e., molecules capable of fixing water at the surface) is promising for heat recovery from industrial processes and solar energy. The typical temperature of in-house warm water systems involving a cogeneration producer does not exceed 63°C, and it can be used for cooling systems and heat pumps. Current processes are based on inorganic porous commercial adsorbents (zeolites or related solids) that suffer from high regeneration temperatures and/or limited pore volumes leading to energy-inefficient systems.
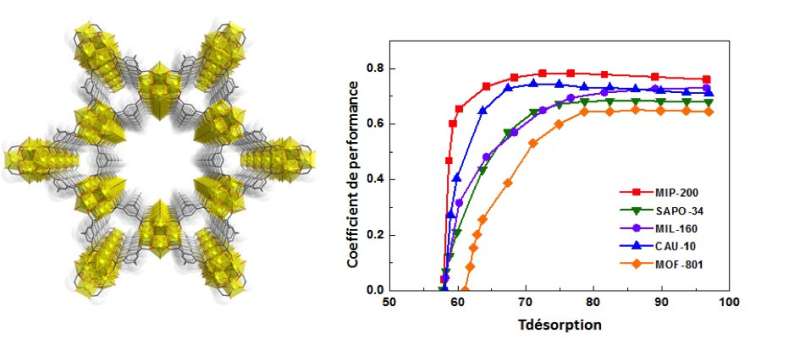
To overcome these drawbacks, researchers from the Paris porous materials Institute and from the Charles Gerhardt Institute in Montpellier designed a new hydrophilic nanoporous hybrid solid with large pores, made of zirconium oxoclusters: Zr-MOF, which combines a set of parameters giving much higher water sorption performance. For cooling processes, the overall performance relies not only on evaporation and condensation temperatures of water, but also on adsorption (exothermic) and desorption (endothermic) temperatures, the storage capacity, stability and kinetics of heat exchange, among others.
The new Zr-MOF exhibits a microporous structure very stable in presence of hot water. It shows a highly pronounced hydrophilic behavior with significant heat exchanges and a pore size sufficient enough to adsorb large amount of water and also a lower regeneration temperature during desorption step (<65°C). Researchers from the Korean Research Institute of Chemical Technology (KRICT) have performed energetic performance calculations (ratio of the energy taken from the evaporator and the energy needed to regenerate the adsorbent). This analysis revealed that the solid is more efficient than any other evaluated porous materials so far for this type of application. It should lead to the development of a new generation of cooling processes to recover solar energy or even energy from heat sources related to human activities.
More information: Sujing Wang et al. A robust large-pore zirconium carboxylate metal–organic framework for energy-efficient water-sorption-driven refrigeration, Nature Energy (2018). DOI: 10.1038/s41560-018-0261-6
Journal information: Nature Energy
Provided by ESPCI Paris