Crystalline material could replace silicon to double efficiency of solar cells
A new material has been shown to have the capability to double the efficiency of solar cells by researchers at Purdue University and the National Renewable Energy Laboratory.
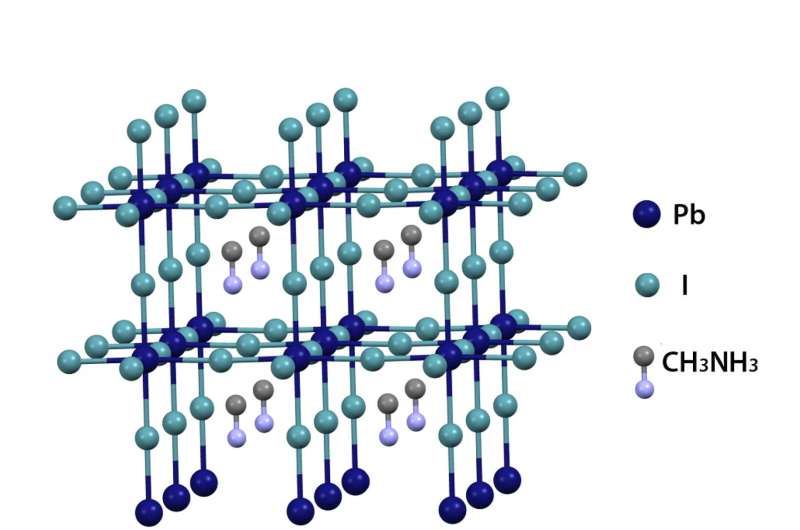
Conventional solar cells are at most one-third efficient, a limit known to scientists as the Shockley-Queisser Limit. The new material, a crystalline structure that contains both inorganic materials (iodine and lead) and an organic material (methyl-ammonium), boosts the efficiency so that it can carry two-thirds of the energy from light without losing as much energy to heat.
In less technical terms, this material could double the amount of electricity produced without a significant cost increase.
Enough solar energy reaches the earth to supply all of the planet's energy needs multiple times over, but capturing that energy has been difficult – as of 2013, only about 1 percent of the world's grid electricity was produced from solar panels.
Libai Huang, assistant professor of chemistry at Purdue, says the new material, called a hybrid perovskites, would create solar cells thinner than conventional silicon solar cells, and is also flexible, cheap and easy to make.
"My graduate students learn how to make it in a few days," she says.
The breakthrough is published this week in the journal Science.
The most common solar cells use silicon as a semiconductor, which can transmit only one-third of the energy because of the band gap, which is the amount of energy needed to boost an electron from a bound state to a conducting state, in which the electrons are able to move, creating electricity.
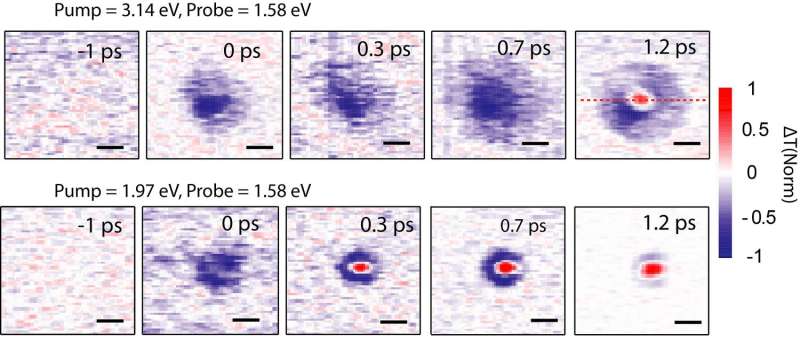
Incoming photons can have more energy than the band gap, and for a very short time – so short it's difficult to imagine – the electrons exist with extra energy. These electrons are called "hot carriers," and in silicon they exist for only one picosecond (which is 10-12 seconds) and only travel a maximum distance of 10 nanometers. At this point the hot carrier electrons give up their energy as heat. This is one of the main reasons for the inefficiency of solar cells.
Huang and her colleagues have developed a new technique that can track the range of the motion and the speed of the hot carriers by using fast lasers and microscopes.
"The distance hot carriers need to migrate is at least the thickness of a solar cell, or about 200 nanometers, which this new perovskite material can achieve," Huang says. "Also these carriers can live for about 100 picoseconds, two orders of magnitude longer than silicon."
Kai Zhu, senior scientist at the National Renewable Energy Laboratory in Golden, Colorado, and one of the journal paper's co-authors, says that these are critical factors for creating a commercial hot-carrier solar cell.
"This study demonstrated that hot carriers in a standard polycrystalline perovskite thin film can travel for a distance that is similar to or longer than the film thickness required to build an efficient perovskite solar cell," he says. "This indicates that the potential for developing hot carrier perovskite solar cell is good."
However, before a commercial product is developed, researchers are trying to use the same techniques developed at Purdue by replacing lead in the material with other, less toxic, metals.
"The next step is to find or develop suitable contact materials or structures with proper energy levels to extract these hot carriers to generate power in the external circuit," Zhu says. "This may not be easy."
More information: "Long-range hot-carrier transport in hybrid perovskites visualized by ultrafast microscopy", Science (2017). science.sciencemag.org/cgi/doi … 1126/science.aam7744
Journal information: Science



















