Correctly packaging the complete yeast genome using purified components in the test-tube
An LMU team has succeeded in correctly packaging the complete yeast genome using purified components in the test-tube. This is a first that yields new insights into the mechanisms of genome organization above the level of the DNA sequence.
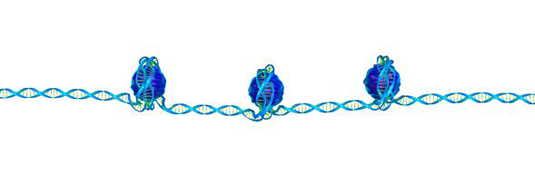
In the cell nucleus, each of the DNA molecules that together comprise the genome (i.e., carry the hereditary information) exists in a compacted form, which is generated by interactions with a specific set of proteins. The resulting DNA-protein complexes are referred to as chromatin. The first level of packaging involves wrapping of DNA around spools made up of four so-called histone proteins. Such wrapped up DNA is referred to as nucleosomes, and their positioning on the DNA plays an important role in the regulation of gene activity—and therefore has a major impact on all cellular processes. Researchers led by PD Dr. Philipp Korber at LMU's Biomedical Center (BMC) have now succeeded in reproducing, in the test-tube, the physiological placement of nucleosomes on the genomic DNA of baker's yeast, starting from purified components. This breakthrough provides fundamental insights into the mechanisms that determine the organization of the DNA in the cell nucleus. The new findings appear in the journal Cell.
To their surprise, Korber and his colleagues discovered that a relatively restricted set of components suffices to ensure proper packaging. In addition to purified yeast DNA and the histone proteins that form the nucleosomes themselves, the only other components essential for the chromatin reconstitution in the test-tube are so-called barrier and remodeler proteins. Barrier proteins serve as alignment points for the nucleosomes, as the LMU team has now directly demonstrated. The experiments have also shed light on the far-reaching importance of the remodeling proteins for the packaging of DNA. "It was already known that remodeling proteins are necessary for nucleosome positioning, but it was unclear whether or not they have a direct and specific role in the process," says Korber. He uses an analogy to explain how these proteins work. Packing goods into a ship's container requires two things – a plan and the people who carry it out. "It was widely thought that remodeler proteins simply carry out orders, doing only what other factors tell them to do. But it turns out that much of the packing plan is built into the remodelers. They bring and follow their own plan during the packing process," says Korber. "In fact, one particular type of remodeler protein acts entirely autonomously, without any input from other factors. That was completely unexpected."
The successful reconstitution of in vivo-like nucleosome positioning on yeast DNA demonstrates that the degree of self-organized DNA packaging is greater than hitherto suspected. Packaging of DNA by remodeler proteins is a dynamic and autonomous process that gives rise to a structure as basis for other essential processes, such as transcription and replication. "This insight into one of the fundamental principles of genome organization is a milestone, and was made possible by the use of carefully controlled conditions for the reconstitution—in the test tube—of the yeast genome as a model system," Korber concludes. "This approach will also facilitate investigations of the role of DNA packaging mechanisms in cancer cells, for chromatin perturbations are known to be directly involved in tumorigenesis."
More information: Genomic Nucleosome Organization Reconstituted with Pure Proteins. Cell. DOI: 10.1016/j.cell.2016.09.045
Journal information: Cell
Provided by Ludwig Maximilian University of Munich