July 3, 2015 report
Radioisotope studies show the continental crust formed 3 billion years ago
(Phys.org)—New research sheds light on how and when the modern day continents began to form. Researchers from the University of Bristol analyzed radio isotope abundances in 13,000 samples of continental crust of varying age and found that the continents began to form around 3 billion years ago. This date may coincide with when plate tectonics began. Their research appears in Nature Geoscience.
One of the difficulties with determining the age of the Earth's crust is that the crust itself is constantly undergoing melting and re-deposition due to subduction. This is a larger factor for the ocean floor than in continental crust because the continental crust is thick and buoyant, making it less susceptible to subduction. By combining information from various radioisotope pairs found in the continental crust, one can unearth clues as to how the crust was formed and when.
In this paper, Dhuime, et al. studied the abundance of rubidium compared to strontium. Rubidium (87Rb) becomes strontium-87 (87Sr) through radioactive decay. Strontium-87 has a half-life of 48.8 billion years. Strontium-86, the most abundant isotope of Sr, is stable and does not undergo radioactive decay. Therefore, the abundance of 87Sr in the continental crust can be traced to the radioactive decay of 87Rb, and based on its half-life, one can determine the age of a rock sample.
Whenever the Earth's crust re-melts or is formed from magma deposition, the amount of 87Rb and silica increases in the residual melt, but the amount of 86Sr does not. Consequently, 87Rb/86Sr correlates with silica content. The silica content is important because the composition of the continental crust is predominantly silica, while the early Earth's crust was likely mafic, or composed of predominantly iron and magnesium.
To calculate 87Rb/86Sr, Dhuime, et al. first determined the 87Sr-to-86Sr ratio and then determined what the ratio was at the time when the crustal melt crystallized. From this they were able to calculate 87Rb/86Sr and therefore determine the amount of silica present at various time periods.
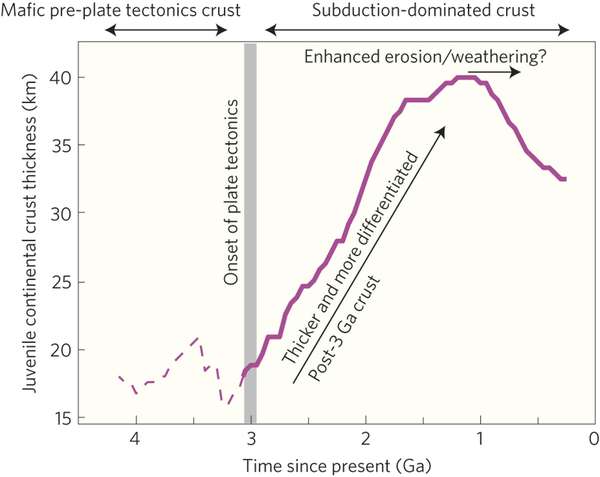
This calculation was performed for over 13,000 volcanic and plutonic rock samples of varying ages, based on neodymium model ages. Results show that at about 3 billion years ago the Earth's continental crust transitioned from a mafic crust to a silica-rich crust. Silica content increased from 3 billion years to 1 billion years ago. At 1 billion years, the amount of silica started to gradually decrease. This result coincides with previous studies using other radioisotope ratios.
Furthermore, Dhuime, et al.'s data show that there is a positive correlation between the increase in 87Rb/86Sr and silica and crustal thickness. The authors point out that the gradual increase of 87Rb/86Sr from 3 billion years to 1 billion years may indicate that the Earth's continental crust was thickening. They estimate that the average thickness of new continental crust increases from ~20 km at 3 billion years ago to ~40 km at 1 billion years ago, and then decreases to ~30 km to the present.
Since subduction is a result of plate activity, the transition at 3 billion years may also indicate the onset of plate tectonics. The thickness of new continental crust reached a maximum and then began decreasing. The authors point out that this may coincide with the time of the Rodinia supercontinent and mountain formation.
Overall, this study provides compelling evidence that the continental crust formed 3 billion years ago, coinciding with the onset of plate tectonics. Additionally, silica content could be used as a metric for determining crust thickness over time.
More information: "Emergence of modern continental crust about 3 billion years ago" Nature Geoscience, 2015. DOI: 10.1038/ngeo2466
Abstract
The continental crust is the principal record of conditions on the Earth during the past 4.4 billion years. However, how the continental crust formed and evolved through time remains highly controversial. In particular, the composition and thickness of juvenile continental crust are unknown. Here we show that Rb/Sr ratios can be used as a proxy for both the silica content and the thickness of the continental crust. We calculate Rb/Sr ratios of the juvenile crust for over 13,000 samples, with Nd model ages ranging from the Hadean to Phanerozoic. The ratios were calculated based on the evolution of Sr isotopes in the period between the TDM Nd model age and the crystallization of the samples analysed. We find that the juvenile crust had a low silica content and was largely mafic in composition during the first 1.5 billion years of Earth's evolution, consistent with magmatism on a pre-plate tectonics planet. About 3 billion years ago, the Rb/Sr ratios of the juvenile continental crust increased, indicating that the newly formed crust became more silica-rich and probably thicker. This transition is in turn linked to the onset of plate tectonics5 and an increase of continental detritus into the oceans.
Journal information: Nature Geoscience
© 2015 Phys.org