June 4, 2015 report
Quality, quantity, and freshness in the reproductive game
(Phys.org)—Many intuitions drawn from our machine world do not smoothly extended to the biological. Whereas the screws or other fasteners used in an automobile typically tend to loosen over time with use, the hardware found in cells tends to tighten with use. This 'use it or lose it' design philosophy applies not only to minds and muscles, but to gonads, perhaps most obviously to the eggs. Although fathers have also come under increasing fire for raising questionable sperm, particularly at age, in many instances the real culprit can be traced simply to sperm age.
While the prolific testes of all species do battle against the ravages of time by mass minting their reproductive minions daily, less obvious are general principles for how unused sperm are eventually laundered and liquidated across species that drastically differ in reproductive strategy. Clelia Gasparini and her colleagues at the University of Western Australia have made some interesting discoveries that bear directly on this question. One thing that their recent Journal of Evolutionary Biology paper clearly shows is that at least in house mice, the familiar industrial economics of quality versus quantity do not necessarily apply.
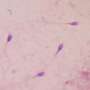
In other words, in looking at mice sourced from populations that varied in their mating frequency and friskiness, those that had the most demanding regimen in terms of sperm production also had the highest sperm quality. While sperm quality may be in a sense, in the eye of the beholder, several expectations repeatedly emerge. We should first note that the researchers didn't quite go as far as tabulating actual DNA mutations, or micromanaging each mating event while accounting for number of sperm per ejaculate. Instead they looked mainly at the total amount of DNA fragmentation—a good general indicator of quality.
At an even more fundamental level of inquiry, the researchers compared mice that were not sexually active for a prior period of two months to those permitted to engage in less virtuous activity. Perhaps unexpectedly, they found that those 'rested males', those deprived of a promiscuous lifestyle, had the same level of sperm quality as the the more wanton mice. The question then, is how do these pent up males avoid the familiar ills of deadly sperm backup (DSB)?
Sperm begin to accumulate damage as soon as they detach from the nourishing Sertoli cells of the testes. After this weaning, particularly long lived sperm would be expected to be further aged after copulation in those species that store them somewhere in the uterine environment prior to fertilization. In many of these species, sperm might expect competition not only among their brethren but from the sperm of other males. One seeming paradox we might observe is that while the great race to the egg is in effect the proving grounds for the oxidative capacity of the respiratory chains of their mitochondria, it is some sense all in vain because in most species male mitochondria are not even passed on.
Unless the male is a mussel or some other creature that does gender in a unique way, then sperm mitochondria either expire like spent salmon, autodestruct, or are subject to active degradation by the egg. This unfortunate compromise appears to be for the greater good. Mitochondria are essentially composites whose protein subunits derive from an optimized match between nuclear and mitochondrial DNA. We have ample evidence, for example, that when embryos are made from heteroplasmic mitochondria (different mitochondria with different mtDNA) then those subunits that make up the electron transport chain don't fit so close anymore and electrons are stalled and detrimentally accumulate at various critical points in the chain.
There is a saving grace to the potential paradox of uniparental inheritance of mitochondria for those hapless mussels we mentioned above: it is preserved by the fact that the males only pass on mitochondria to their (male) sons. There are clues to the resolution to the other paradox, namely the question of why bother selecting for fast or powerful sperm mitochondria when those mitochondria never see the light at the end of tunnel. For one, sperm need to do more than just sprint to the egg, they also have to get in, and stop the others from making an unsightly polyspermically fertilized egg.
At this point we might need to leave terra firma in order to imagine how the rested mice or other species avoid DSB and compromised sperm. Hypothesized mechanisms include degradation in the testes, spontaneous ejaculations, self-relief, or transient modifications to the plumbing so that any surfeit of sperm spills into the urine. One problem here, is that all of these mechanisms require some fairly sophisticated programming.
Take for example, nocturnal emissions in humans. That they occur is not up for debate, but how and why they occur most surely is. Did their evolution single-handedly create our capacity to dream, perhaps directly or alternatively as a side-effect? There is no reason to stop there either. If dreams, then why not some of the other idiosyncratic nuances we associate with high level visuo-ideation or even consciousness itself. Food for thought for another day, but perhaps a bit much to pin on the need for gamete freshness. More pressingly, if we are to fully account for the massive and ongoing sperm production across species with both frequent and infrequent mating, we might need to hypothesize further mechanisms beyond quality control to explain it.
One idea has emerged from recent and controversial investigations into Lamarckian inheritance of ancestral fears. In particular, the idea has been floated that if sperm are the agents responsible for the transmission of an acquired characteristic, then they must be doing more than just transmitting static nuclear DNA. Therefore, if information regarding the current state of the organism and their environment is deposited into sperm in an ongoing fashion, then we already have some appreciation both for the needs of liberality and freshness.
It will no doubt be critical to do similar sperm quality studies on other species and assess the broadly cast concept of 'Fertilization outcome' in each of them. Sperm not only come in endless forms most beautiful in different species but they are not always doomed to decay with the predictable delay of the petals of a tulip. It is obviously more than sperm quantity and quality that controls what can only be seen as a cooperative event between the sexes.Accommodations made for the finiteness of sperm by females include tailoring their ovulation, particular under reproductively stressed times to the dynamics of male supply. Examples here would be cyclic ovulation on seasonal, monthly, or other scales supplemented by various degrees reflex or facultative ovulation, or in the extreme scarcity of males, virgin birth or parthenogenesis.
More information: Sexual rest and post-meiotic sperm ageing in house mice, Journal of Evolutionary Biology, DOI: 10.1111/jeb.12661
Abstract
Fertilization by aged sperm can result in adverse fitness consequences for both males and females. Sperm storage during male sexual rest could provide an environment for post-meiotic sperm senescence causing a deterioration of the quality of stored sperm, possibly impacting on both sperm performance (e.g. swimming ability) and DNA quality. Here, we compared the proportion of sperm with fragmented DNA, an indicator of structural damage of DNA within the sperm cell, among males that had been sexually rested for approximately two months, to that of males that had mated recently. We found no evidence of intra-epididymal sperm DNA damage or any impairment in sperm performance, and consequently no evidence of post-meiotic sperm senescence. Our results suggest that male house mice are likely to possess mechanisms that function to ensure that their sperm reserves remain stocked with "young", viable sperm during periods of sexual inactivity. We also discuss the possibility that our experimental design lead to no difference in the age of sperm among males from the two mating treatments. Post-meiotic sperm senescence is especially relevant under sperm competition. Thus, we sourced mice from populations that differed in their levels of postcopulatory sexual selection, enabling us to gain insight into how selection for higher sperm production influences the rate of sperm ageing and levels of DNA fragmentation. We found that males from the population that produced the highest number of sperm also had the smallest proportion of DNA-fragmented sperm, and discuss this outcome in relation to selection acting upon males to ensure that they produce ejaculates with high quality sperm that are successful in achieving fertilizations under competitive conditions.
Journal information: Journal of Evolutionary Biology
© 2015 Phys.org