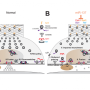
How Microprocessor precisely initiates miRNA production
A scientific group from the Center for RNA Research within the Institute for Basic Science (IBS) and School of Biological Sciences in Seoul National University has reported an insightful molecular mechanism of how Microprocessor, the DROSHA-DGCR8 complex, precisely determines cleavage sites on miRNA-containing primary transcripts allowing faithful initiation of microRNA biogenesis.
The group's findings, published in Cell on 28th May as Advance Online Publication, not only reveal the function of each part of human Microprocessor, but also outline future work on the molecular structure of the protein complex which will enable various new applications of RNA interference technology in basic research and human therapeutics.
MicroRNAs (miRNAs) are short RNA species of ~22 nucleotides, but play critical roles in a wide variety of cellular processes, including stem cell differentiation and tumorigenesis. Their gene silencing mechanism is dependent on their sequences which are acquired through the miRNA production process, called miRNA biogenesis. This process is initiated in the nucleus by Microprocessor, a complex of the catalytic subunit DROSHA and a co-factor DGCR8, which cleaves primary transcripts (pri-miRNAs) containing miRNA sequences. It is thought that the pri-miRNA processing step determines the sequences of miRNAs and thereby their actions, thus it is greatly important to understand how Microprocessor accurately process pri-miRNAs.
A group of researchers in IBS and SNU led by Dr. Jae-Sung Woo and Dr. V. Narry Kim have made a significant advance toward understanding the molecular mechanism of pri-miRNA processing by using highly pure recombinant Microprocessor which was lacking in this field. They discovered that Microprocessor consists of one DROSHA and two DGCR8 molecules. They also disclosed an important and surprising role of DROSHA as a "molecular ruler" by showing that DROSHA can recognize the ssRNA-dsRNA junction at the lower side of pri-miRNA and measure the distance of ~11 base pairs from the junction to find the precise cleavage sites. DROSHA was also found to specifically recognize the UG motif located at the lower junction, allowing it to interact with pri-miRNAs more specifically. Over the previous knowledge, DGCR8 was found to have three functionally distinct parts: the tail to stabilize DROSHA, the body to enhance the processing efficiency by recruiting pri-miRNA, and the head to ensure the processing accuracy by recognizing the upper elements of pri-miRNA including the apical UGU motif.
Dr. Nguyen T. A., the first author of this paper, has integrated various biochemical, biophysical, and bioinformatical data and proposed a current model showing that the functional parts of Microprocessor interact with the cis-acting elements on pri-miRNA for accurate processing. This model also represents an interesting molecular mechanism of the Microprocessor orientation on pri-miRNAs which has been unknown so far. Furthermore, it brings out a comprehensive understanding how Microprocessor acts differently on various pri-miRNA substrates with different sequence and structural features, and clarify decade-standing controversies over the pri-miRNA processing mechanism.
More information: Nguyen, T. A., Jo, M. H., Choi, Y.-G., Park J., Kwon, S. C., Hohng, S., Kim, V. N.* and Woo, J.-S.* (2015) "Functional anatomy of the human Microprocessor" Cell, in press, (*co-corresponding authors), DOI: dx.doi.org/10.1016/j.cell.2015.05.010
Journal information: Cell
Provided by Institute for Basic Science