Bright like a diamond: lasers and compressed carbon recreate Jupiter's core

While missions like the Kepler can tell us quite a bit about other worlds, but to actually look into the heart of a planet we had to put a diamond through a pretty rough road-test.
Last week a team of scientists at the Lawrence Livermore National Laboratory (LLNL) announced that they recreated the pressures found in Jupiter's core by applying consistently increasing the level of compression on a sample of carbon. This technique - called ramp compression - provided a first look at the material states that dwell under intense interior-planetary pressures.
"If you understand the properties of materials deep within the planetary interior you can get a better picture of [planetary] evolution," physicist Ray Smith LLNL told Astrobio, "Understanding the properties of the constituent elements helps interpret external observational data ."
The observations to which Smith is referring are those made by Kepler and other similar missions, which are discovering planets around other stars with increasing frequency. Under certain circumstances, like during transits, these observations can reveal details such as the radius and mass of an exoplanet. However, no observations have directly revealed what's inside an exoplanet.
Instead, Smith told Astrobio, we rely on models. These model are based on the mass-radius relationship, which we can get from Kepler, but they use assumptions that we haven't been to able to verify, until now.
"Observations from the Kepler mission are limited because the planets are so far away," said Smith. "Scientists can determine the Mass-Radius of an exoplanet and can then speculate on the composition-assuming its comprised of a homogeneous material." In other words, in order to fit the observations to the models, exoplants are sometimes assumed to be 100% gaseous, or 100% diamond, or 100% made of water.
Clearly, without taking a closer look, we know that exoplanets are not 100% water. Planets are not 100% any single substance in any single physical state. Because of changes in pressure, materials near the center of planets exist in different states than the materials on the outside. This is true for Earth, for Jupiter - whose core is something of a mystery - and for planets of all sizes in other solar systems.

So the question becomes: how can we verify our models of the structure of exoplanets, or even our own planet?
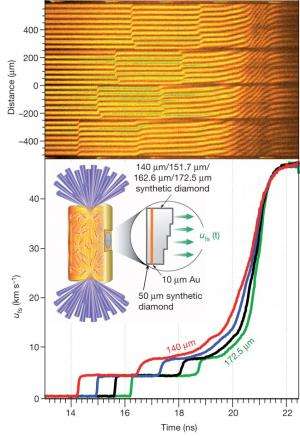
The answer offered by Smith, Gilbert Collins and their collaborators at the National Ignition Laboratory includes a facility the size of 3 football fields, 176 laser beams, a tiny gold tube and a very small diamond.
While diamond may be precious to us on Earth, it is likely to exist in abundance in the Universe at large. Like the graphite lying the center of every #2 pencil, diamond is simply a stable derivative of carbon. As the fourth most abundant element in the Universe, carbon also comprises the cores of many planets, including, probably, some of our own gas giants.
In order to discover the state of carbon and other elements inside of planets like Jupiter and Saturn, Smith and colleagues bombarded a tiny carbon sample with x-rays - a maneuver that compressed the carbon but didn't cause it to heat rapidly. The result was a cool diamond under more than 50 million atmospheres of pressure, just like the carbon in Jupiter's core.
While diamonds may not literally lie at the heart of Jupiter (though some have proposed that they do), the stable carbon structure worth a fortune on Earth may well make up the interior of Neptune - a planet whose blue-green atmosphere is mostly methane. At sea-level on Earth, or one atmosphere, methane is a gas made of one carbon and four hydrogens. So near the core of Neptune we would expect to find abundant carbon. At high pressures-millions of atmospheres-those carbon atoms would arrange themselves into a diamond structure, and then perhaps into something beyond diamonds.
"At progressively higher pressures, the carbon atoms will change their configuration," said Smith. "At over 10 million atmospheres of pressure, the carbon atoms are predicted to rearrange themselves so they would no longer be in a diamond structure, but rather they will assume a different [higher density] structural arrangement. It will still be carbon, but, so the theory predicts, the crystal structure and the mechanical and chemical properties will be different."
Neptune-sized planets comprise the bulk of the 1000+ currently confirmed and potential exoplanets. So that theoretical beyond-diamond substance, while new to us, might also be extremely common.
When Smith and colleagues retrieve and analyze their sample, they may be able to say for sure what kind of carbon exists beyond diamonds. Whether or not that substance has a previously unseen structure, Smith and colleagues now claim that they have fashioned a window into the core of the most common kind of planet in the Galaxy.
"We've been relying on models," said Smith. "For the first time we are actually able to measure in these pressure ranges what the material density really is."
In the future, the team at the LLNL plans to repeat these crushing experiments in an attempt to re-create the structure of the key substance in Earth's core: iron.
"We've already taken shots on iron," Smith said. "And plan to extend this research to a range of other cosmologically important materials."
Provided by Astrobio.net
This story is republished courtesy of NASA's Astrobiology Magazine. Explore the Earth and beyond at www.astrobio.net .