A salt-free primordial soup?

Most scientists who study the origin of life assume that it occurred in the ocean. But a minority view is that ions in seawater may interfere with prebiotic chemistry, making a freshwater environment more likely.
The saltiness of our blood is often cited as evidence that life originated in the ocean. However, some researchers contend that the first chemical steps toward biology would have been easier in freshwater rather than saltwater.
The exact location for the origin of life is still a wide open question, but many scientists have assumed that it happened somewhere in the ocean.
"The main argument for a marine origin is that there is so much seawater," says David Deamer of UC Santa Cruz. Roughly 98% of the Earth's water bodies are salty, and this percentage was likely much higher 4 billion years ago when we think the first life-forms made their appearance.
But Deamer doesn't think quantity is a substitute for quality. Seawater, in his estimation, is too reactive with certain biomolecules to have served as the "broth" for the primordial soup.
A freshwater origin seems to have been what Charles Darwin was proposing when he imagined the spontaneous formation of biomolecules in "some warm little pond."
Deamer and his colleagues are testing Darwin's idea, but with the temperature turned up. They have gone to several geothermally heated "ponds" around the world to see if they can't cook up some of the more complex molecules of life in these freshwater environments. Deamer recounts these adventures in a new book called "First Life: Discovering the Connections between Stars, Cells, and How Life Began."
His critics might say the work could use a pinch of salt.
The ocean in your veins
It's no accident that our blood is about a quarter as salty as the ocean. This level is tightly regulated by the kidneys. Our cells will die if the salt level in blood and other fluids goes too high or too low.
The normal salt that we are familiar with is sodium chloride (NaCl). In solution, the salt breaks up into ions: specifically positively-charged sodium ions and negatively charged chloride ions. All cells – human and otherwise – spend a great deal of time shuffling these and other ions around. This shuffling is necessary to maintain the fluid pressure inside the cell, but it also creates electric potentials that provide a kind of "battery" for performing certain cellular functions.
"This sort of bioenergy is common to all life forms," says Shiladitya DasSarma of the University of Maryland Biotechnology Institute.
The ubiquity of ion-mediated potentials in cells may be telling us something about where life got started.
"I wouldn't think ions could play such an important role unless they were around in the beginning," he says.
DasSarma believes that the first organisms arose in salt water that was perhaps extremely salty. The early ocean was perhaps twice as salty as it is today. Moreover, the ingredients of life may have been concentrated by evaporation in a seaside pool or lagoon, which would have concentrated the salt as well.
Bursting life's bubble
The problem with seawater, according to Deamer, isn't the salt, per se. Seawater also contains other ions, like those of magnesium and calcium, which carry a charge of +2. These so-called divalent ions react unfavorably with certain building blocks of life.
For example, calcium ions readily bind with phosphate, thus making this molecule unavailable for important biological functions, such as energy transfer (in the case of adenosine triphosphate, or ATP) and genetic coding (as part of the backbone of DNA and RNA).
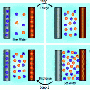
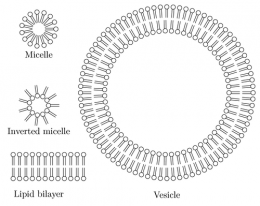
Deamer is especially concerned with the effect that divalent ions have on simple fatty acids. These "soapy" molecules – generically called lipids – line up together to form closed vesicles. Several scientists have theorized that self-forming "bubbles" of this sort might have served as a kind of rudimentary cell membrane for the very first organisms.
However, the simple vesicles can't form in seawater because the divalent ions react with the fatty acids. People with mineral-rich "hard" water in their homes are familiar with this chemistry. Soap products don't lather as well with hard water, which has high concentrations of calcium and magnesium ions that react with the soap molecules to form a solid that we call soap scum.
"Seawater would definitely precipitate fatty acids, preventing membrane formation," says Jack Szostak of Harvard University. "So I agree with Dave Deamer that primitive cells had to live in a fresh water environment. "
Throwing the catalyst out with the seawater
The challenge for Deamer is that those divalent ions are far from a nuisance when it comes to other aspects of biochemistry.
DasSarma points out that divalent magnesium ions are needed for important phosphate chemistry, and calcium ions play a vital role in cellular signaling.
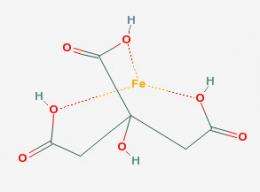
Moreover, "some of those divalent ions are transition metals, which I think of as being involved with ligands in pre-macromolecular catalysis," says Harold Morowitz of George Mason University.
Transition metals are elements (like iron, manganese and nickel) that occupy the middle of the Periodic Table. They trade electrons fairly easily, which makes them good catalysts for driving chemical reactions.
When transition metals combine with small organic molecules called "ligands," they can drive important chemical reactions. Nowadays, this catalysis is done by proteins, but these large molecules are so complex that it's hard to imagine them being around at the dawn of life. Morowitz believes transition metals were necessary to get the biological ball rolling.
Michael Russell from the Jet Propulsion Lab seems to agree: "It is the inorganic elements that bring organic chemistry to life." And he goes on to stress that these elements can only stay in solution in saltwater (with its abundant chloride ions), otherwise they tend to precipitate into solids where they no longer can play their biological roles.
Contrary to Deamer's position, Russell doesn't believe life necessarily needed a lipid vesicle in the beginning. He thinks the prebiotic chemistry could have begun inside tiny pores of rocks. Here, proteins and DNA could have assembled in a closed environment.
"It's the proteins that do the work," Russell says. "The lipids are merely the castle wall."
Membranes-first
Deamer doesn't deny that some of the first biological steps may have occurred inside pores or on the surface of clay minerals. But eventually, organisms freed themselves from these fixed structures and ventured out into open water. And that's when they would need a good "container."
"At some point during its origin, life started using membranes," Deamer says.
As any fish will tell you, there are ways to make membranes that are "salt-proof," but these are complicated structures that need to be synthesized by enzymes or something similar, says Deamer. The far easier route is to use spontaneously forming membranes that work great in freshwater. Even though divalent ions would be scarce in this environment, the first proto-cells could still probably scavenge some if needed.
"I certainly would not claim that life began in distilled water," Deamer says. He believes life would need some ions to get going. "It's just that seawater is too much of a good thing."
So where might early life have found a nice freshwater launching pad? The Earth had no continents 4 billion years ago, as the planet was essentially one big ocean. But geologists believe that there were volcanic islands, like Hawaii and Iceland, which could have trapped fresh rain water in ponds or lakes.
Szostak believes these freshwater bodies could accumulate useful organic molecules (in contrast to the ocean where everything tends to get diluted). Being near volcanoes could have provided heat for creating wet-dry cycles. Experiments have shown that these cycles can concentrate lipid molecules to help them organize into membranes.
Deamer has witnessed first-hand these wet-dry cycles in ponds next to modern-day volcanoes in Kamchatka, Hawaii and California. He and his colleagues went so far as to dump lipid molecules into the ponds to see if they might form membranes "in the wild." The answer was no. The organic material attached itself to clay minerals at the bottom of the ponds (something that wouldn't have likely been a problem on the early Earth).
But these field tests haven't deterred Deamer.
"I've learned from visiting these places what to do to simulate these environments in the lab," he says.
His team has built a "hot pond" simulator. Little vials with freshwater and the basic ingredients of life are heated to above 60 degrees Celsius and routinely re-wetted with "rain water" from a syringe. Recent results have shown that membrane-forming lipids not only form vesicles, but they may help drive DNA replication -- something that modern cells need protein enzymes to do.
All the simulations have so far used freshwater, but Deamer says they plan to test saltwater to see how the results change.
The salt habit is hard to break.
Source: Astrobio.net