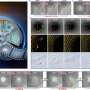
Carving at the nanoscale
Researchers at the Catalan Institute of Nanotechnology have successfully demonstrated a new method for producing a wide variety of complex hollow nanoparticles. The work, published this week in Science, applies well known processes of corrosion in a novel manner to produce highly complex cage-like nanoscale structures with potential applications in fields from medicine to industrial processing.
A common theme in nanoscience research is the recycling of "old" processes and protocols that were once applied crudely on bulk materials in trades and industrial settings, but which can now be applied to nano-sized structures with high precision and resolution using newly available instruments and know-how.
After several years of research, Spanish scientists at the Catalan Institute of Nanotechnology (ICN) have refined methods based on traditional corrosion techniques (the Kirkendall effect and galvanic, pitting, etching and de-alloying corrosion processes). They show that these methods, which are far more aggressive at the nanoscale than in bulk materials due to the higher surface area of nanostructures, provide interesting pathways for the production of new and exotic materials.
By making simple changes in the chemical environment it is possible to tightly control the reaction and diffusion processes at room temperatures, allowing for high yields and high consistency in form and structure. This should make the processes particularly attractive for commercial applications as they are easily adapted to industrial scales.
A wide range of structures can be formed, including open boxes, bimetallic and trimetallic double-walled open boxes with pores, multiwalled/multichamber boxes, double-walled, porous and multichamber nanotubes, nanoframes, noble metal fullerenes, and others.
Asides from their intrinsic beauty, such nanostructures present new options for drug delivery, catalysis, remediation of contaminants and even structural components for nanoscale robots.
More information: "Carving at the Nanoscale: Sequential Galvanic Exchange and Kirkendall Growth at Room Temperature", doi:10.1126/science.1212822
Provided by Institut Catala de Nanotecnologia