January 6, 2011 feature
Scientists make holograms of atoms using electrons
(PhysOrg.com) -- While holography is often associated with artistic 3D images, it can also be used for many other purposes. In a new study, scientists have created holograms of atoms using laser-driven electron motion, which could lead to a new type of ultra-fast photoelectron spectroscopy. In the future, this type of holography could enable scientists to study the structures of molecules in a more direct way than before.
The scientists, Ymkje Huismans from the FOM-Institute AMOLF in Amsterdam, The Netherlands, and an international research team have published their study in a recent issue of Science Express.
“What we have experimentally demonstrated is that it is possible to make holograms by taking an electron out of a molecule and, using a laser field, redirect the electron toward the molecule,” coauthor Marc Vrakking, of the FOM-Institute AMOLF and the Max Born Institute in Berlin, told PhysOrg.com.
In their experiments, the scientists beamed an intense infrared laser light at an atom or molecule, which resulted in the atom or molecule becoming ionized and releasing an electron. The laser field causes the liberated electron to oscillate away from and toward the ion. Sometimes, an electron and ion collide, releasing a very short burst of radiation.
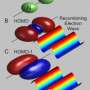
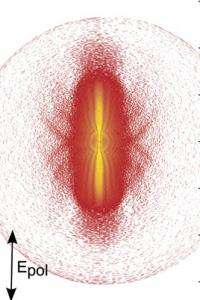
Because the electron motion is fully coherent, meaning it always has the same phase, the scientists realized that they could apply holographic techniques to record information about the ion and electron. The key to holographic electron imaging is to observe the interference between a reference wave (which is emitted by the electron and doesn’t interact with the ion) and a signal wave (which scatters off the ion and encodes its structure). When the reference wave and signal wave interfere on a detector, the encoded information about the electron and ion is stored and can be viewed in the future. As the scientists explained, the result is a hologram of an atom produced by its own electrons.
The researchers also developed theoretical models to simulate their measurements, confirming that the hologram had stored spatial and temporal information about the electrons and ions. By using the holographic structures to develop a new kind of ultra-fast photoelectron spectroscopy, researchers could be able to directly measure electron and ion movements on the attosecond timescale. This ability would be useful for understanding chemical reactions at the most basic level, particularly in molecules that cannot be easily studied by other methods.
More information: Y. Huismans, et al. “Time-Resolved Holography with Photoelectrons.” Science Express. 16 December 2010. DOI:10.1126/science.1198450
Copyright 2010 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.