Great balls of evolution: Microbiologists evolve microorganisms to cooperate in new way
University of Massachusetts Amherst microbiologists Derek Lovley, Zarath Summers and colleagues report in the Dec. 2 issue of Science that they have discovered a new cooperative behavior in anaerobic bacteria, known as interspecies electron transfer, that could have important implications for the global carbon cycle and bioenergy.
The scientists found that microorganisms of different species, in this case two Geobacter species, can form direct electrical connections and pass an electric current from one microbe to the other. By cooperating in this way the two microbes can consume food that neither of them could use on their own.
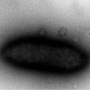
The cell aggregates or "great balls of evolution" that Summers evolved in the laboratory look very much like those found in nature which are involved in degrading organic matter into the greenhouse gases, carbon dioxide and methane. Conversion of wastes to methane by microbial aggregates is an increasingly popular method for producing natural gas as an renewable energy source.
Others can be found consuming methane from vents at the bottom of the ocean. In both cases, investigators have been puzzled for years about how these aggregates function, because a 40-year-old interspecies hydrogen transfer paradigm did not seem to fit observations. Now, the mystery appears to be solved.
As Lovley, the principal investestigator, explains, "We placed the microbes under conditions in which they had to work together in order to survive and grow using the alcohol we gave them as an energy source. They're the ultimate drinking buddies, collaborating to consume ethanol." With support from the Genomic Science Program of the U.S. Department of Energy, his lab has been exploiting the ability of microorganisms to adapt to novel conditions and developing microbes for practical applications.
It's been known since the 1960s that microorganisms can indirectly exchange electrons by the process known as interspecies hydrogen transfer. In it, one microbe produces hydrogen that another microbe then consumes. It was experiments carrried out by doctoral candidate Summers to explore this phenomenon further that led to discovery of the new direct transfer process.
To begin, Summers put two species of Geobacter together under conditions expected to favor hydrogen-sharing interactions. At first, the cells did cooperate to consume the alcohol by sharing hydrogen. Over time, they also started clumping together and transforming the culture from one of dispersed microscopic cells, invisible to the naked eye, to a collection of complex multi-cellular structures, millimeters in diameter.
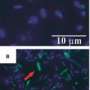
Resisting her lab mates' urgings to shake the cultures and break up the unexpected cell clumps, Summers continued to allow the spheres to grow. Now they were exhibiting a deep red color due to the presence of iron-containing proteins known as cytochromes. When observed with an electron microscope, they had clearly developed an intricate structure with a series of channels, presumably to help nutrients enter. They had also established completely new electric connections that permitted them to directly share electrons.
"The direct electron transfer is much more efficient and they consume alcohol much faster this way," Summers points out. Sequencing the DNA in the big red balls revealed the secret to this electrical connection: a mutation in one of the Geobacter species had caused it to make much more of a cytochrome known as OmcS. Previous studies in Lovely's lab had shown that OmcS lines up along Geobacter's electrically conductive filaments known as microbial nanowires.
"This turn of events suggested that the cytochrome was key to the electrical connection between the cells" says Summers. This was confirmed in subsequent experiments with genetically manipulated microbes. When the researchers deleted genes for the cytochrome or the nanowires, the microbes did not form the red balls and never effectively used their alcohol fuel. Lovley, Summers and colleagues had thus pinpointed the source of the microbes' new behavior.
Further experiments showed that if the mutation was introduced before putting the two Geobacters together, they rapidly formed the balls and consumed alcohol. Deleting a gene that would be necessary for the cells to exchange hydrogen also hastened ball formation, demonstrating that interspecies hydrogen transfer was not an important factor. "This is a clear case of life evolving to function more effectively in a new environment" says Lovley.
"We're guessing that many types of natural aggregates rely on interspecies electron transfer" said Lovley. "We already have some good preliminary evidence for this with some more complex natural systems. With DNA sequencing we can determine how the microbes evolve when challenged to do better. We can learn a lot about the basic mechanisms of the process of interest," he adds.
Provided by University of Massachusetts at Amherst