Reprogramming the debate: stem-cell finding alters ethical controversy
When University of Wisconsin-Madison researchers succeeded in reprogramming skin cells to behave like embryonic stem cells, they also began to redefine the political and ethical dynamics of the stem-cell debate, a leading bioethicist says.
R. Alta Charo, a UW-Madison professor of law and bioethics, says the scientific finding could have far-reaching effects on the social dimensions of the ongoing controversy over embryonic stem cell research.
"This is a method for creating a stem cell line without ever having to work through, at any stage, an entity that is a viable embryo," Charo says. "Therefore, you manage to avoid many of those debates with the right-to life community."
The research alters the debates surrounding both human embryonic stem cell research and somatic cell nuclear transfer cloning.
For ordinary embryonic stem cell research, it offers a means of obtaining pluripotent cell lines from a non-embryonic source. For cloning research, it offers a means to make customized, pluripotent cell lines without having to create an intermediate embryo that is a "clone" of an adult person, she says.
Charo says the discovery could remove objections from critics on both the right and left wings of the political spectrum.
To derive embryonic stem cells, it is necessary to remove critical cells from an embryo, resulting in its destruction. That triggers opposition from right-to-life critics of the research, who cite moral and ethical concerns. The research has also generated opposition from some members of the women's movement who object the use of stimulating drugs in women who agree to donate eggs for cloning research aimed at creating specialized embryonic stem cell lines.
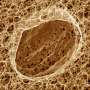
The latest findings have the potential to render both of those objections moot, since the research showed that introducing four genes into cells derived from skin cells, called human fibroblasts, resulted in cells that essentially share all the features of embryonic stem cells - but without using or destroying embryos.
"It holds a great deal of promise for freeing this whole area of research from those two main sources of friction," Charo says.
The discovery also creates questions about the future of government funding for traditional embryonic stem cell research, which in recent years has been a contentious political issue.
"It's going to fuel those who call for preferential federal funding only for non-embryonic stem cell research and it will certainly complicate any efforts to expand funding for embryonic stem cell research at the federal level," she says.
Twice, Congress has passed legislation to overturn the Bush administration policy on embryonic stem cell research and allow funding for research using any embryonic stem cell lines, not just those designated by the administration in 2001. Twice, the president has vetoed it.
"Any piece of research like this that suggests that we can get cell lines that are equally usable without having to go through an embryo in intermediate steps is going to undermine any effort on the part of Congress to overturn the Bush policy," she says.
The latest discovery, however, is likely to suggest avenues of research that are already eligible for federal funding. Recently, the White House directed the National Institutes of Health to emphasize the funding of research that examines alternative means for obtaining pluripotent cell lines whose usefulness is comparable to that of human embryonic stem cells.
"No matter how well this new technique can be used for many of the disease-research and disease-treatment applications foreseen for embryonic stem cell and cloning research, however, calls for criminalization or wholesale de-funding of embryonic stem cell and cloning research are not warranted," Charo adds. "Criminalizing any area of science, as opposed to merely regulating it, would be contrary to the political and constitutional traditions of academic and scientific freedom, as well as the historical spirit of inquiry that characterizes this country."
Charo serves on several expert advisory boards of organizations with an interest in stem cell research, including CuresNow, the Juvenile Diabetes Research Foundation, the International Society for Stem Cell Research and WiCell, as well as on the advisory board to the Wisconsin Stem Cell Research Program.
In 2005, she was appointed to the ethics standards working group of the California Institute for Regenerative Medicine. Also in 2005, she helped to draft the National Academies' Guidelines for Embryonic Stem Cell Research, and in 2006 she was appointed to co-chair the National Academies' Human Embryonic Stem Cell Research Advisory Committee.
Source: University of Wisconsin-Madison