Anthrax vaccine produces immunity with nanoparticles, not needles
A vaccine against anthrax that is more effective and easier to administer than the present vaccine has proved highly effective in tests in mice and guinea pigs, report University of Michigan Medical School scientists in the August issue of Infection and Immunity.
The scientists were able to trigger a strong immune response by treating the inside of the animals’ noses with a “nanoemulsion” – a suspension of water, soybean oil, alcohol and surfactant emulsified to create droplets of only 200 to 300 nanometers in size. It would take about 265 of the droplets lined up side by side to equal the width of a human hair.
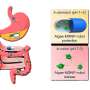
The oil particles are small enough to ferry a key anthrax protein inside the nasal membranes, allowing immune-system cells to react to the protein and initiate a protective immune response. That primes the immune system to promptly fight off infection when it encounters the whole microbe.
Besides eliminating the need for needles, the nanoemulsion anthrax vaccine has another advantage, the researchers say: It is easy to store and use in places where refrigeration is not available.
An effective and easy-to-administer vaccine would be a valuable tool for health authorities dealing with any future attack in which a terrorist might spread anthrax microbes. The researchers say a nasal nanoemulsion-based anthrax vaccine, if it proves effective in humans, could be given easily to people even after they are exposed in an anthrax attack, along with antibiotics. With some diseases, vaccines given after exposure are used to boost the speed of the immune response.
“Anthrax spores can remain in the environment or even in the lungs of exposed individuals for some time. Nasal vaccination could be given to build up immunity after anthrax exposure and improve the outcome of other treatments,” says Anna Bielinska, Ph.D., the paper’s lead author and a scientist at the Michigan Nanotechnology Institute for Medicine and Biological Sciences at the U-M. She is also an assistant research professor in the Allergy Division of Internal Medicine at the U-M Medical School.
In the 2001 attack in which anthrax spores were mailed to members of Congress and reporters, some anthrax-exposed individuals were given antibiotics but not the existing anthrax vaccine, because it requires six injections over a period of six months and may cause adverse reactions.
In the new study, the U-M team combined the nanoemulsion and a recombinant protein of Bacillus anthracis to make the vaccine, which they gave first to mice in either one or two applications. They found the animals developed several types of effective immune response. The vaccine produced both systemic and cellular immunity, meaning that the body produces antibodies and primes specific cells throughout the body to fend off anthrax infection. The vaccine also induced immunity on the mucous membranes of the nose and lungs, where inhaled anthrax spores enter the body and start a process that can cause illness and death.
“We saw protective immunity in the animals after only two administrations rather than six,” says James R. Baker, Jr., M.D., director of the Michigan Nanotechnology Institute for Medicine and Biological Sciences and the senior author of the study. He is the Ruth Dow Doan Professor in the Department of Internal Medicine and chief of the Division of Allergy and Clinical Immunology in the U-M Medical School.
After administering the vaccine, the researchers challenged the immune systems of immunized guinea pigs with injections of 1,000 times the lethal dose of Bacillus anthracis spores. All the animals survived, whereas none of the control animals did.
When the researchers delivered large doses of Bacillus anthracis spores directly into the animals’ nasal tissue, they found that 40 percent to 70 percent of the immunized animals survived.
Because of the biosafety restrictions on the use of anthrax, these challenge experiments were done at the Batelle Memorial Institute and the University of Texas Medical Branch, which have labs federally approved for handling the pathogen.
Source: University of Michigan Health System