March 30, 2007 feature
New Nanomaterial, 'NanoBuds,' Combines Fullerenes and Nanotubes
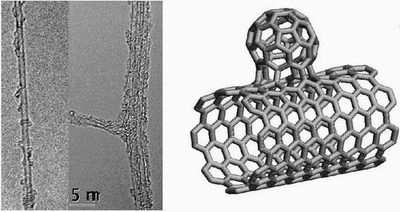
Researchers have created a hybrid carbon nanomaterial that merges single-walled carbon nanotubes and spherical carbon-atom cages called fullerenes. The new structures, dubbed NanoBuds because they resemble buds sprouting on branches, may possess properties that are superior to fullerenes and nanotubes alone. They are described in the March 2007 edition of Nature Nanotechnology.
“Both fullerenes and single-walled carbon nanotubes exhibit many advantageous properties, but despite their similarities there have been very few attempts to physically merge them. The novel hybrid material we discovered merges the two into a single structure, in which the fullerenes are covalently bonded to the nanotubes,” said Esko Kauppinen, a scientist involved in the work, to PhysOrg.com. Kauppinen is a professor and researcher at the Helsinki University of Technology and the technology development organization VTT Biotechnology, both in Finland.
Synthesis of the NanoBuds began with the synthesis of single-walled carbon nanotubes (SWNTs) in a standard reactor. The resulting SWNTs seemed to be coated with clusters of carbon atoms, but a closer investigation, using an electron microscope, revealed that most of the clusters were actually fullerenes.
The fullerenes coating the nanotubes displayed unusual behavior. Using a transmission electron microscope, the researchers saw that these fullerenes did not move around upon the nanotube surface. This in not typical of fullerenes on SWNTs and indicated a strong fullerene/nanotube bond, which was further verified by several tests.
The group further probed the NanoBuds' structure using ultraviolet spectroscopy. The resulting UV spectra showed the attached fullerenes to be similar to C70, the 70-carbon-atom fullerene, which has a slightly ellipsoidal shape compared to C60 and other spherical fullerenes. This deviation from the characteristic fullerene sphere, or “bucky ball,” could have been due to the presence of covalently attached oxygen or hydrogen. Further measurements detected oxygen in each NanoBud, and a round of infrared spectroscopy revealed the presence of two types organic compounds known as ethers and esters. These compounds may act as bridge-like structures connecting the fullerenes to the nanotubes.
Kauppinen and his colleagues say that NanoBuds may find use as cold electron field emitters – materials that emit electrons at room temperature under a high applied electric field – due to the fullerenes' many curved surfaces, which make for better emitters that flat surfaces. Cold electron field emission is key to many technologies, including flat-panel displays and electron microscopes.
“We believe that NanoBuds may have other applications, such as molecular anchors to prevent SWNTs from slipping within composite materials,” says Kauppinen. “Additionally, since the optical and electrical properties of the fullerenes and nanotubes can be individually tuned, NanoBuds provide SWNTs with distinct regions of different electrical properties. This could be useful for many applications, including memory devices and quantum dots.”
Citation: Albert G. Nasibulin, Peter V. Pikhitsa, Hua Jiang, David P. Brown, Arkady V. Krasheninnikov, Anton S. Anisimov, Paula Queipo, Anna Moisala, David Gonzalez, Günther Lientschnig, Abdou Hassanien, Sergey D. Shandakov, Giulio Lolli, Daniel E. Resasco, Mansoo Choi, David Tománek and Esko I. Kauppinen, “A novel hybrid carbon material.” Nature Nanotechnology 2, 156-161 (2007)
Copyright 2007 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.