Physicists discover structures of gold nanoclusters
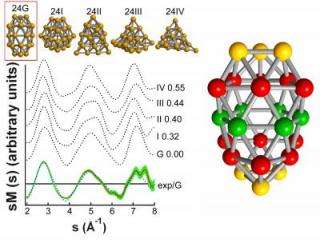
Using different experimental techniques, two separate and independent research groups in collaboration with a team from the Center for Computational Materials Science (CCMS) at the Georgia Institute of Technology, have unveiled the size-dependent evolution of structural and electronic structural motifs of gold nanoclusters ranging in size from 11 to 24 atoms. The experiments, in conjunction with the theoretical analysis performed by the Georgia Tech team, show near perfect agreement pertaining to the cluster structures occurring in the experiments.
Understanding the electronic and geometric structures of gold nanoclusters is a key step towards understanding their behavior under different conditions, such as their use as nanocatalysts or in certain medical applications. The results appear in separate papers in The Physical Review B and in the journal ChemPhysChem.
In its bulk form, gold is treasured for its property as a non-reactive metal. Its use in electronics, dentistry, jewelry and art, depends on this inertness. But at the nano scale, when gold clusters contain only a small number of atoms, gold shows very different properties, which exhibit chemical reactivity that make them potent catalysts. Because their chemical and physical properties depend greatly on their physical structures, significant efforts have been invested by scientists to determine what the most stable configurations of gold clusters are in this size range. Understanding this is of great importance for elucidating the chemical properties of these clusters and in research aiming to discover the physical patterns that govern how the clusters are put together.
Between 2000 and 2002, a Georgia Tech team, led by Uzi Landman, director of CCMS, Regents' and Institute professor, and Callaway chair of physics at Georgia Tech, predicted that negatively charged gold nanoclusters, up to 13 atoms in size, would exhibit two-dimensional, flat structures. The appearance of two-dimensional structures for such relatively large metal clusters is unique to gold, and the researchers showed that it is related to the strong relativistic effects for this metal. When these predictions were verified experimentally, research in Landman's group and in other places focused on what happens when the nanoclusters are even larger.
"We wanted to know, what happens after 13 atoms," said Landman. "What happens when these clusters become three-dimensional and what is their structural motif?" For the past few years, scientists at the CCMS have made theoretical predictions about the structures of gold nanoclusters in the larger size range. Now, working with two independent experimental groups, Landman and his collaborators have found firm evidence pertaining to the size-dependent structural development of these nanoclusters.
One of these collaborations involved researchers from the University of Freiburg and the Fraunhofer Institute for Mechanics of Materials, both in Germany, and a scientist from the University of Jväskylä in Finland. The Freiburg team performed photoemission experiments, in which a laser is shot at the gas-phase cluster causing it to eject an electron. Measuring the energy profiles of the emitted electrons using lasers of different wavelengths allowed the researchers to gain knowledge about the occupied electronic energy levels in the clusters. The distribution of these levels depends on the specific geometric arrangement of atoms in the clusters. Indeed, the theoretical analysis of the correlation between the distributions of the electronic energy levels and the atomic spatial arrangements allowed the researchers to determine the clusters' electronic properties, as well as geometric structures.
In the other collaboration, the Georgia Tech researchers worked with a team from the Rowland Institute at Harvard University. They used electron diffraction, a technique in which a beam of electrons is fired at the clusters, causing the electrons to scatter. By measuring the intensity of the scattered electrons and comparing it to the change in momenta of the electrons caused by their collisions with the atoms of the clusters, they obtaines information about the spatial arrangements of the atoms in the clusters. Theoretical analysis of the interference patterns in these measured intensities allowed them to determine the clusters' structures.
"It turns out that close to all the stable structures that were found through our theoretical analysis of the photoemission measurements were the same as those that emerged from analysis of the electron scattering experiments," said Landman. "In our analysis we have used first-principles electronic structure calculations based on density-functional theory, in conjunction with structural optimization techniques. This is likely the first time that two separate and independent experimental tools, in conjunction with a common theoretical analysis, have shown such a high degree of agreement in the challenging area of structural determination of nano clusters."
To avoid any bias, and ensure that the groups' analyses weren't being unintentionally influenced by knowledge of each other, neither experimental group saw the results of the other until the publication of their respective papers.
The results of the Georgia Tech collaborative investigations with the European group are published in the journal ChemPhysChem Volume 8, (2007), and those obtained from the collaboration with the Rowland Institute are published in The Physical Review B volume 74, (2006).
Through this comparison between experiment and theory, the teams found that the clusters start out as two-dimensional structures till 13 or 14 atoms in size, changing to three-dimensional hollow cages from about 16 atoms, and developing a face-centered-cubic tetrahedral structure at 20 atoms, resembling the bulk gold crystalline structure. However, at 24 atoms the gold clusters take an unexpected capped tubular cigar shape.
"These results assist us not only in determining the structures of the clusters, but also provide insight into the factors that underlie their self-assembly," said Landman. "In some ways, we are determining the 'structural grammar' of these gold nanoclusters and by understanding that, we may better understand what motifs appear as we continue to search for the structures of clusters larger than 24 atoms.
Source: Georgia Institute of Technology