Research Helps Uncover the Secrets of an Age-Old Killer
Scientists working in part at the Stanford Synchrotron Radiation Laboratory (SSRL) have discovered a gene for a protein that regulates the cellular response to copper in the bacterium that causes tuberculosis. Copper is a biologically essential element, but its levels within a cell must be carefully controlled because too much can cause cell death. These findings, reported in the January issue of Nature Chemical Biology, explain how a wide variety of bacteria control copper concentrations within their cells, and this understanding could lead to new treatments for tuberculosis.
The research team, led by David Giedroc of Texas A&M University, discovered the gene that encodes a "Copper-sensitive operon Repressor" (CsoR), which controls the production of copper-binding proteins and is present in many types of bacteria.
"Metal ions like copper are the 'Achilles heel' of tuberculosis bacilli and other pathogenic organisms," said Giedroc. "Too much of these ions inside a cell is deadly, but the cell needs them to break down reactive compounds that would otherwise destroy important proteins, DNA, and lipids within the cell."
Copper ions are prevented from damaging the cell by regulatory proteins that sense the metal and turn on the production of other proteins that help mitigate the deadly effects of copper. However, the gene responsible for turning on these proteins and the mechanism behind how the protein works had not been previously identified in many bacteria.
The researchers hypothesized that the gene that encodes the copper-binding protein must appear somewhere in the genome of Mycobacterium tuberculosis, the pathogen responsible for tuberculosis infections in humans. The University of Wisconsin's Adel Talaat, a co-author on the study, had previously shown that CsoR was part of a cluster of genes active in M. tuberculosis infecting the lungs of mice. Analysis of the DNA sequence of another nearby gene led the researchers to hypothesize that it encoded a protein that acts as a "copper pump" that drives excess copper out of the cell, and that CsoR was the critical regulator of this process.
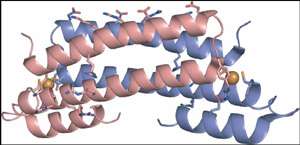
Using the synchrotron facilities at SSRL and the Canadian Light Source (CLS), the researchers used a technique called x-ray absorption spectroscopy (XAS) on purified samples of the copper-binding protein from Mycobacterium tuberculosis. With the XAS technique, a beam of x-rays is passed through a sample, exciting the electrons in the sample's copper atoms, thereby revealing important clues about sample's chemistry and how its atoms are bonded. Using this and other techniques, Giedroc and his team uncovered the chemical mechanism behind the copper-binding protein in M. tuberculosis and thus how the protein functions.
"It's a fundamental new discovery which might, down the road, lead to new treatments for tuberculosis," said University of Saskatchewan's Graham George, who collaborated with Giedroc's team on the experiment. "The x-ray absorption spectroscopy part of the work was vital to understanding how the CsoR protein binds copper."
Another member of Giedroc's team, James Sacchettini of Texas A&M, employed a commonly used technique called x-ray crystallography to help determine the overall structure of the copper-binding protein. With this technique, protein molecules are grown into crystals and then exposed to a beam of x-rays. By measuring the pattern of x-rays scattered by the sample, researchers can map out the arrangement of the atoms in a molecule and gather clues about its function.
Read the full paper in Nature Chemical Biology here.
Source: by Brad Plummer, SLAC

















