Sediments in northern Gulf of Mexico not right for methane gas hydrate formation
Marine sediments in the northern Gulf of Mexico are likely too warm and salty to hold the amount of methane gas hydrates – a potential energy resource – originally thought to exist in the ocean floor there.
Researchers reported high-resolution geophysical and geochemical data for two representative sites off the coast of New Orleans, La., that suggest previous estimates for the region should be revised sharply downward, according to their paper published in the March 15 issue of the journal Geophysical Research Letters (GRL).
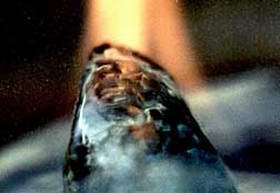
Image: A gas hydrate sample synthesized in a Georgia Tech research lab from tetrahydrofuran can sustain a flame as gas is released from the structure during heating. Photo courtesy Carlos Santamarina
“We found that conditions are not favorable for the formation of methane gas hydrates at these sites because of the geology of the northern Gulf of Mexico, which consists of salt domes that one can think of as mushroom clouds of salt that rise buoyantly through sediments,” said Carolyn Ruppel, an associate professor of geophysics in the Georgia Institute of Technology’s School of Earth and Atmospheric Sciences and lead author on the paper. “The thermal properties of salt make the sediments hotter there, and the heat, coupled with the presence of the salt in pore spaces, makes it harder to form gas hydrates.”
The research, a collaboration between Georgia Tech, Rice University and the Scripps Institution of Oceanography, was funded by the National Science Foundation. The researchers continue to analyze their data to get a quantitative estimate of the gas hydrates at these sites, but the deposits are likely thin or non-existent, Ruppel said. Researchers collected their data during a two-week research cruise in October 2002.
Methane gas hydrates are touted as a potential new source of natural gas, but scientists are also studying them because they may contribute to global warming and could represent a threat to deep-sea petroleum production.
Methane is produced by the decomposition of organic material in the sediment or by thermal processes similar to those responsible for the formation of oil. As the methane moves through the sediment, it combines with water at the low temperatures and high pressures beneath the ocean to produce an ice-like solid. Methane gas hydrates exist along continental margins worldwide, most in sediments tens to hundreds of meters below the sea floor in waters more than 500 meters deep. These hydrates exist as disseminated deposits, chunks several centimeters across and sometimes as concentrated layers.
In the northern Gulf of Mexico, previous research on potential methane gas hydrates assumed homogeneous conditions (e.g., same temperature and geology) and did not consider the impact of salt on hydrate formation, Ruppel noted.
“The methods we used are very good at helping us understand the conditions in the sediment and make a prediction of what’s there,” she explained. “We found conditions that are not compatible with published estimates that imply large methane gas hydrate deposits in the northern Gulf.”
Ruppel and her colleagues took a multidisciplinary approach, using overlapping methods to characterize the two sites they studied, she said. They used high-resolution seismic equipment from the lab of co-author and Georgia Tech Assistant Professor Daniel Lizarralde to image the sea floor and to find conduits through which fluids could flow.
Geochemist and co-author Gerald Dickens of Rice University worked with graduate students to analyze water samples from cores of sediment extracted from the sea floor. They developed chemical profiles that revealed, for example, salt and sulfate concentrations. Sulfate measurements are important for understanding the biology of the system, specifically the interaction of microbes that produce sulfate and methane.
Ruppel was responsible for high-resolution heat flow measurements to constrain temperature and the rate of fluid flow in the sediments. Collaborators from Scripps Institution of Oceanography also collected data on fluid flux from the sea floor in the northern Gulf – an important constraint on the hydrology of the system and its potential for hydrate formation, Ruppel noted. These measurements were not incorporated into the analysis published in GRL.
“When you put all of this together, you get a good idea of the conditions in the gas hydrate reservoir – that is, the sediments that contain gas hydrates,” Ruppel added.
Research to characterize methane gas hydrates is ongoing throughout North America and the world, Ruppel noted. She and her colleagues have also studied hydrates at Blake Ridge off the South Carolina coast during normal oceanographic cruises, deep sea drilling legs, and submersible dives. On the Blake Ridge, they characterized hydrates both below and on the ocean floor. Through the international Ocean Drilling Program, other scientists have drilled or will soon drill boreholes to explore hydrates off the coasts of Oregon and Vancouver.
Other research is focused on hydrates as potential energy sources, as well as the safety issues related to drilling. These issues include the potential for sea floor destabilization that could occur as hot fluids are pumped up from deep sediments through the hydrate stability zone, Ruppel explained. “Methane gas hydrates are like ice,” she added. “They can melt and cause the sea floor to collapse. The oil companies are interested in understanding this issue better.”
Ruppel and colleague J. Carlos Santamarina, a Georgia Tech professor in the School of Civil and Environmental Engineering, will focus on these questions when their research group participates in drilling this spring in the northern Gulf of Mexico. The U.S. Department of Energy is funding this work through a joint industry project with ChevronTexaco.
“There’s a lot of research on hydrates going on,” Ruppel said. “Ultimately, these studies around North America and the world will shed more light on how much hydrate is out there. I hope that will get us closer to answering the question about whether hydrates are a viable energy resource. It’s going to take some time. If we do learn it’s a viable resource, then we’ll have to face a new set of issues on how to actually produce energy from this resource.”
Producing methane from gas hydrates faces some daunting challenges. A key question is whether it would take more energy to extract the gas hydrates than the gas may provide, Ruppel added.
Source: Georgia Institute of Technology















