Open Microfluidic and Nanofluidic Systems
The labs of the future will be "labs-on-a-chip", i.e., integrated chemical and biochemical laboratories shrunk down to the size of a computer chip. An essential prerequisite for such labs are appropriate microcompartments for the confinement of very small amounts of liquids and chemical reagents. Directly accessible surface channels, which can be fabricated by available photolithographic methods, represent an appealing design principle for such microcompartments and, thus, provide a new route towards open microfluidic and nanofluidic systems. Scientists from the Max Planck Institute of Colloids and Interfaces, the Max Planck Institute of Dynamics and Selforganization and the University of California in Santa Barbara have shown that such open systems are possible in general but only if the geometry of the surface channels is carefully matched with their wettability (PNAS 102, 1848-1852 (2005).
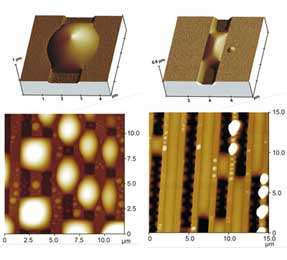
Fig.1: Atomic (or scanning) force microscopy images of liquid morphologies on silicon substrates with rectangular surface channels which have a width of about one micrometer. On the left, the liquid does not enter the channels but forms large lemon-shaped droplets overlying the channels (dark stripes). On the right, the liquid enters the channels and forms extended filaments separated by essentially empty channel segments (dark stripes). In the bottom row, several parallel surface channels can be seen in both images; in the top row, there is only one such channel with a single droplet (left) or filament (right). Close inspection of the upper right image reveals (i) that this filament is connected to thin wedges along the lower channel corners and (ii) that the contact line bounding the meniscus of the filament is pinned to the upper channel edges.
Many research groups around the world work towards the construction of "labs-on-a-chip" in order to integrate chemical and biochemical analyzers on the micrometer or even nanometer scale. These devices will significantly change the way in which research is performed in the life sciences since they offer the ability to work with much smaller reagent volumes, much shorter reaction times, and the possibility of massive parallel processing. In general, this should lead to increased throughput and, thus, to reduced cost of (bio)chemical analysis. In addition, such integrated labs-on-a-chip have many potential applications in biomedicine and bioengineering. In the context of biomedicine, for example, they could provide fast and detailed analysis of blood samples in the physician's office without the need to wait several days before the sample has been returned from specialized laboratories. Other applications include customized chips for space travel in order to monitor microbes inside spacecraft or to detect life on other planets.
An obvious prerequisite for such miniaturized labs are appropriate microcompartments for the confinement of very small amounts of liquids and chemical reagents. Like the test-tubes in macroscopic laboratories, these microcompartments should have some basic properties: They should have a well-defined geometry by which one can measure the precise amount of liquid contained in them; they should be able to confine variable amounts of liquid; and they should be accessible in such a way that one can add and extract liquid in a convenient manner.
An appealing design principle for such microcompartments is based on open and, thus, directly accessible surface channels which can be fabricated on solid substrates using available photolithographic methods. The simplest channel geometry which can be produced in this way corresponds to channels with a rectangular cross section. The width and depth of these channels can be varied between a hundred nanometer and a couple of micrometer.
At first sight, it seems rather obvious to use such surface channels as microcompartments. However, if one actually tries to fill these channels with a certain liquid, one observes that the liquid often refuses to enter the channels. In fact, as shown in the new PNAS study, liquids at surface channels can attain a large variety of different wetting morphologies including localized droplets, extended filaments, and thin wedges at the lower channel corners. Examples for these morphologies as observed by atomic (or scanning) force microscopy (AFM) are shown in Figure 1.
When the AFM experiments were first performed, it was not known how to produce a certain liquid morphology since there was no systematic theory for the dependence of this morphology on the materials properties and on the channel design. Such a theory has now been developed. This theory addresses the strong capillary forces between substrate material and liquid and takes the ‘freedom’ of contact angles at pinned contact lines into account. Such a contact line is visible in the upper right image in Figure 1. In such a situation, the contact angle is not determined by the classical Young equation but can vary over a wide range of values.
A surprising prediction of the new theory is that the experimentally observed polymorphism of the wetting liquid depends only on two parameters: (i) the channel geometry, i.e., the ratio of the channel depth to the channel width; and (ii) the interaction between substrate material and liquid. One has to distinguish seven different liquid morphologies which involve localized droplets (D), extended filaments (F), and thin wedges (W) at the channel corners. For microfluidics applications, the most important morphology regime is (F) which corresponds to stable filaments. Since this regime covers a relatively small region of the morphology diagram, it can only be obtained if one carefully matches the channel geometry with the substrate wettability. Thus, a water filament in a narrow channel that has a width of 100 nanometer can sustain an overpressure up to 15 atm. In contrast, if the channel had a width of one millimeter, the water filament could only sustain a thousandth part of an atmosphere.
One relatively simple application of the morphology is obtained if the system is designed in such a way that one can vary or switch the contact angle in a controlled fashion. One such method is provided by electrowetting; alternative methods, which have recently been developed, are substrate surfaces covered by molecular monolayers that can be switched by light, temperature, or electric potential.
The experiments described in the PNAS study use a polymeric liquid that freezes quickly and can then be scanned directly with the tip of an atomic force microsope. However, the same morphology diagram should also apply to other liquids and other substrate materials. It should also remain valid if one further shrinks the surface channels and, in this way, moves deeper into the nanoregime. As one reaches a channel width of about 30 nanometer, one theoretically expects new effects arising from the line tension of the contact line, but such nanochannels have not been studied experimentally so far.
The new PNAS study provides an instructive example for the close relation between basic research and technological development in the micro- and nanoregime: open systems with directly accessible surface channels can be used for micro- and nanofluidic applications but only if one carefully matches the channel geometry with the substrate wettability. This constraint is a direct consequence of the strong capillary forces that dominate in the micro- and nanoregime and can be formulated in a quantitative way using the methods of theoretical physics. In general, the development of any new technology requires a systematic understanding of the underlying physics. This latter constraint applies to all length scales: if one wanted to build a robot which walks over water, for instance, a human-like robot is a bad idea while a spider-like robot is a much better choice.
Source: Max Planck Institute of Colloids and Interfaces















