Finding the right mix: A biomaterial blend library
From dental implants to hip replacements, biomaterials have become big business. But scientists pursuing this modern medical revolution share a basic challenge: biocompatibility. How will a biomaterial on the lab bench actually work inside the human body? Will a patient accept the new material or suffer an inflammatory response? And can that material survive in a human's complex system?
To tackle such questions, researchers at the National Institute of Standards and Technology (NIST) and the New Jersey Center for Biomaterials (NJCB) at Rutgers University have developed new methods to analyze the interactions between cells and biomaterials. Their work could lead to inexpensive techniques for building better biomaterials.
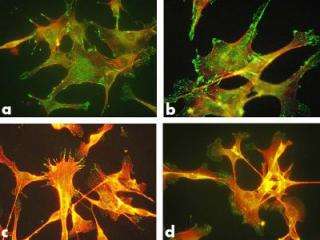
Polymers derived from the amino acid tyrosine make up a broad class of degradable biomaterials under investigation. Such materials provide a temporary scaffold for cells to grow and tissue to regenerate. In a 2006 study presented at the national meeting of the American Chemical Society in September, the researchers analyzed how two types of model cells--immune cells known as macrophages and bone cells known as osteoblasts--responded to changes in the composition of thin films made of these tyrosine-derived polymers. In practice, many biomaterials are made from blends of polymers to achieve specific material properties.
Optimizing the blend composition is often a difficult and time-consuming task. As the blends gained a higher or lower proportion of a respective material, the cells around them react by changing shape, ultimately increasing or decreasing contact with the films. In the body, such cell-material dynamics are critically important to the outcome--determining whether a biomaterial leads to inflammation or abnormal cell growth, for example.
The new study represents an innovative line of research. Working with NJCB, NIST scientists have developed a method for constructing "scaffold libraries" --collections of biomaterial scaffolds made from controlled polymer blend compositions. The library currently contains scaffolds made from blends of poly(DTE carbonate) and poly(DTO carbonate). Ultimately, Becker says, the goal is to develop rapid, inexpensive methods to predict the behavior in the body of any of thousands of possible tyrosine-derived blends.
Citation: L.O. Bailey, M.L. Becker et al. Cellular responses to phase-separated blends of tyrosine-derived polycarbonates. Journal of Biomed. Mater. Res. Part A. March 1, 2006.
Source: NIST