Scientists Work on Nanowires That Could Fuel Molecular Technology
Before nanotechnology can deliver on its promise of molecular-sized machinery and super-charged computer chips, scientists must still produce the most basic of components: nano-sized wire.
But if chemistry professors at the University of Wisconsin-Milwaukee are successful, they could help turn the concept of nanotechnology into reality. Chemistry professors Dennis Bennett and Wilfred Tysoe have been working on building insulating, highly conductive "nanowires," the necessary first step in developing circuitry at the atomic scale.
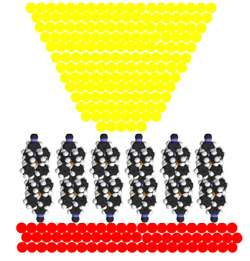
Image: A schematic illustrating the method for measuring current in nanowires.
People have been talking for a long time about molecular wires," says Bennett. "In fact, scientists have already been able to construct such wires, but they are relatively short and must be isolated from their neighbors by immersing them in a sea of other, non-conducting molecules." A series of molecules that Bennett has developed are unique because they have the potential to yield insulated nano-scale wires.
Bennett and Tysoe are collaborating with researchers at Northwestern and Iowa State universities
in a project submitted to the National Science Foundation to understand the forces that govern the atomic universe, including the nature of conductivity at nanoscale.
Nanotechnology, or nanoscience, refers to the study of objects in the size range of nanometers. A nanometer is about 10 times the size of a hydrogen atom. For example, if you were to line up objects at this scale, you would be able to fit over a billion of them in one inch, says Tysoe. This offers the possibility, for example, of placing trillions of miniscule devices on the area of a dime.
But at this small scale, conditions can be very different from those of the classical physical environment. It makes sense, says Tysoe, because a single atom will behave differently from a bulk piece of material containing trillions and trillions of atoms.
"There's a lot of talk about it, but relatively little work focuses on the regime of the transition between the classical physical world and the quantum world. That's where the science comes in," says Bennett.
The diameter of a single E. coli bacterium is as small as a wire can get before it stops acting classically, says Bennett.
In their collaborative project, Bennett designs the molecules and Tysoe studies the nature of surface interactions and their conductive properties. They believe they have the right molecule and the right metal surface. The next step will be connecting the two.
They also are measuring the electrical properties of Bennett's molecules to see if their properties are similar to those of large-scale wires.
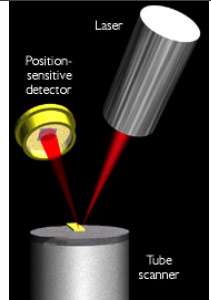
It is a delicate process. With forces much less than those exerted by the foot of an ant, a small gold tip is brought into contact with the surface, a voltage is applied, and the current is measured through an array of nanowires (see illustrations at right).
"We are finding that, although the molecules do conduct extremely well, the current that passes through them is not directly proportional to the voltage applied, the way it would be with bulk wires," says Tysoe.
Another potential application for this class of molecules is as tunable catalysts.
A catalyst is a substance that can increase the rate of a chemical reaction by lowering the energy required to form a product. Catalysts are used in a myriad of ways, such as to make useful products from crude oil, minimize harmful emission from automobiles, or produce fertilizers.
Their efficacy, however, generally depends on the electronic structure of their components. Since the metals in the nanowires molecule can be electrically connected to a surface, this offers the possibility of adjusting its electronic properties.
"This means we can envisage tuning the properties of the catalyst merely by applying a voltage, and even of having the same catalyst do several different jobs," says Bennett.
A host of ultra-sensitive instrumentation is aiding the progress in nanoscience.
"What's new is that we are able to ‘see' the components of atoms and molecules and measure their properties," says Tysoe, "using various microscopic techniques such as scanning tunneling microscopy (STM), transmission electron microscopy, and electron diffraction."
These tools are currently in use at UWM, in the laboratories of Lian Li and Marjia Gajdardziska-Josifovska. The two physics professors – along with Bennett, Tysoe and seven other professors – are members of UWM's interdisciplinary Laboratory for Surface Studies.
For the foreseeable future, Bennett and Tysoe are committed to a long, careful process of experimentation and testing.
There are few breakthrough moments, says Bennett, only a steady accumulation and refinement of knowledge that can lead to ideas. "Science is the creative engine of technology," he says. "The research leads you to it, not the other way around."
Source: University of Wisconsin-Milwaukee


















