Researchers describe how cells take out the trash to prevent disease
Garbage collectors are important for removing trash; without them waste accumulates and can quickly become a health hazard. Similarly, individual cells that make up such biological organisms as humans also have sophisticated methods for managing waste.
For example, cells have developed complex systems for recycling, reusing and disposing of damaged, nonfunctional waste proteins. When such systems malfunction and these proteins accumulate, they can become toxic, resulting in many diseases, including Alzheimer's, cystic fibrosis and developmental disorders.
Scott Emr, director of the Weill Institute for Cell and Molecular Biology at Cornell, and colleagues, describe in detail how cells recycle protein waste in two recent papers appearing in the journals Cell and Developmental Cell.
"We are interested in understanding how cells deal with garbage," said Emr. "It's really a very sophisticated recycling system."
Cells use enzymes known as proteases to break down proteins into their component amino acids in the cytoplasm -- the fluid inside the cell's surface membrane. Those amino acids are then reused to make new proteins. But water-insoluble proteins embedded in the cell's membrane require a much more complicated recycling process.
Emr's paper in Cell identifies a family of proteins that controls the removal of unwanted water-insoluble proteins from the membrane. The research advances understanding of how cells recognize which proteins out of hundreds on a cell's surface should be removed. For example, hormone receptors at a cell's surface signal such processes within the cell as growth and proliferation. To inactivate these receptors and terminate the growth signal, receptors are tagged for removal. Failure to inactivate can lead to developmental diseases and cancer.
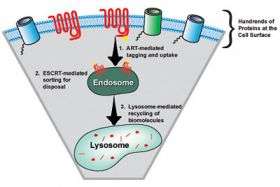
The researchers, including postdoctoral fellows Jason MacGurn and Chris Stefan, identified nine related proteins in yeast, which they named the "arrestin-related trafficking" adaptors or ARTs. Each of these proteins identifies and binds to a different set of membrane proteins. Once bound, the ART protein links to an enzyme that attaches a chemical tag for that protein's removal. The ARTs are found in both yeast and humans, suggesting the fundamental nature of their function.
Once the protein is tagged, the piece of membrane with the targeted protein forms a packet, called a vesicle, that enters the cell's cytoplasm. There, the vesicle enters a larger membrane body called an endosome, which in turn dumps it into another compartment called the lysosome, where special enzymes break apart big molecules to their core units: proteins to amino acids, membranes to fatty acids, carbohydrates to sugars and nucleic acids to nucleotides, and those basic materials are then reused.
The paper in Developmental Cell, co-authored by Emr with postdoctoral fellows David Teis and Suraj Saksena, describes for the first time how a set of four proteins assemble into a highly ordered complex. This complex encircles membrane proteins that must be disposed of in the lysosome. Emr's lab was the first to identify and characterize these protein complexes (known as ESCRTs). The Developmental Cell paper describes the order of events in which the ESCRT complexes encircle and deliver "waste" proteins into vesicles destined for recycling in the lysosome.
Emr's ESCRT discovery has allowed researchers to better understand how the AIDS virus is released from its host cell. HIV hijacks the cell's ESCRT machinery during virus budding. "So, if you block the function of ESCRTs, you could block HIV release," said Emr.
Provided by Cornell University
















