Scientists identify a molecule that coordinates the movement of cells
Even cells commute. To get from their birthplace to their work site, they sequentially attach to and detach from an elaborate track of exceptionally strong proteins known as the extracellular matrix. Now, in research to appear in the October 3 issue of Cell, scientists at the Howard Hughes Medical Institute and Rockefeller University show that a molecule, called ACF7, helps regulate and power this movement from the inside - findings that could have implications for understanding how cancer cells metastasize.
"The most dangerous part of cancer is that cancer cells migrate from their primary location and invade other parts of the body," says first author Xiaoyang Wu, a postdoc in Elaine Fuchs's Laboratory of Mammalian Cell Biology and Development. "ACF7 facilitates cell movement, so it's possible that the less ACF7 a cell has, the less malignant it would become. It's a really exciting question in cancer biology now."
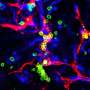
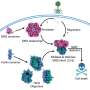
To travel along the extracellular matrix, cells must stick to and unstick from it via focal adhesions, structures composed of molecules that connect the inside to the outside of the cell. (While some molecules connect to the matrix, others connect to a scaffold inside the cell called the cytoskeleton.) As these structures collectively assemble and disassemble, the cell walks forward. Fuchs and Wu show that ACF7 can not only access energy stores to power this movement from within but also coordinate it by linking two fiber-like proteins called f-actin and microtubules, which together form the cytoskeleton and help give cells their shape.
"Inside the cell, actin cables converge at focal adhesions at the cell's leading edge," Fuchs explains. "We found that ACF7 guides microtubules along a roadway of actin cables and leads them toward the focal adhesions at the cell's periphery. Among the cargo transported along microtubules are factors that disassemble focal adhesions. Hence by coupling microtubule, actin and focal adhesion dynamics within the cell, ACF7 becomes an orchestrator of directed cellular movement."
In particular, Wu and Fuchs, who is also a Howard Hughes Medical Institute investigator and Rebecca C. Lancefield Professor at Rockefeller, found that without ACF7, microtubules were no longer guided toward the focal adhesions in a directed manner. They also noticed that cellular movement slowed, suggesting that the sticky adhesive sites were no longer assembling and disassembling efficiently.
To figure out why, Fuchs and Wu studied how quickly wounds heal in mice. "During injury, stem cells proliferate and migrate to the affected site and replenish lost cells," explains Wu. "We saw that the cells without ACF7 proliferated normally, but they moved very, very slowly compared to normal skin cells. So the problem wasn't with abnormal proliferation but with cell migration." When the researchers mutated ACF7 so it couldn't release stored energy in cells, ACF7 linked f-actin and microtubules but the cells were also sluggish in their movement.
In previous work, the Fuchs team had already showed that ACF7 appeared side by side with focal adhesion molecules, but they never knew, until now, that ACF7 guides microtubules along actin cables to these sites. "Now, we have a better idea of why it's important for ACF7 to be there," says Fuchs. "In order to make the adhesive sites dynamically stick and unstick, assembly and disassembly factors need to be recruited there. The intracellular roadway governed by ACF7 makes that possible."
In the future, this information could be relevant in developing cancer therapeutics. "A major goal in the clinical arena is to halt cancer cells from migrating, a process important in metastasis," says Fuchs. By suppressing ACF7's function in cancer cells, it might be possible to slow metastasis.
Source: Rockefeller University