Catalysis discovery takes aim at NOx emissions
A discovery in molecular chemistry may help remove a barrier to widespread use of diesel and other fuel-efficient "lean burn" vehicle engines. Researchers at the Department of Energy's Pacific Northwest National Laboratory have recorded the first observations of how certain catalyst materials used in emission control devices are constructed.
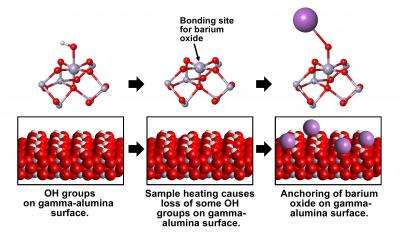
The PNNL team observed how barium oxide attaches itself to the surface of gamma-alumina. Barium oxide is a compound that absorbs toxic nitrogen oxide, commonly referred to as NOx, from tail-pipe emissions. Gamma alumina is a form of aluminum oxide that is used as a support for catalyst materials, such as barium oxide, that are the active ingredients in exhaust systems.
"The discovery is encouraging because understanding catalysts in molecular and atomic detail can directly identify new ways to improve them," said PNNL researcher Janos Szanyi. The manner in which barium oxide anchors onto alumina suggests the exact site where catalytic materials begin to form - and where they can be available to absorb NOx emissions.
Lean burn engines deliver up to 35 percent better fuel economy because they mix more air with gasoline than standard internal combustion engines. But the more efficient engines can't meet strict emissions standards because current aftertreatment devices don't effectively reduce NOx emissions. New catalysts are essential before the economic and environmental benefits of lean burn engines can be realized.
Alumina is a common and relatively inexpensive catalyst support material. Its surface structure, formation and thermal stability have been the subjects of much research, but the alumina particles are too small and poorly crystalline for traditional surface analysis. Researchers used the world's first 900-MHz nuclear magnetic resonance spectrometer to reveal the anchoring behavior. The instrument is located at the William R. Wiley Environmental Molecular Sciences Laboratory, a DOE national scientific user facility at PNNL.
Scientists know that the aluminum ions in alumina coordinate, or bond, to either four or six oxygen ions. When water is present, 10 to 15 percent of the aluminum ions on the surface bond to six oxygen ions: one underneath to the bulk of the alumina, four in a square on the surface and one on top to an oxygen ion in the water molecule.
Removing the water by heating leaves the aluminum ion with only five oxygen bonds. In this "penta-coordinated" state, the aluminum is open for bonding to the barium oxide. Results from the NMR spectrometer showed that the catalyst filled every available penta-coordinated site, atom-for-atom.
The team is now examining the interaction of gamma-alumina with other metal and metal oxide particles to determine if penta-coordinated aluminum ions are suitable bonding locations for other catalytic materials.
Reference: Ja Hun Kwak, Jain Zhi Hu, Do Heui Kim, Janos Szanyi and Charles Peden. "Penta-coordinated Al3+ Ions as Preferential Nucleation Sites for BaO on ã-Al2O3." Journal of Catalysis 251(1):189-194. July 2007.
Source: Pacific Northwest National Laboratory















