Two proteins found on telomeres control DNA damage response pathways
No sooner had cells evolved linear chromosomes than they had a life-threatening problem to solve. To the machinery that repairs broken DNA, chromosome ends look a lot like breaks in need of mending, so they could elicit a DNA damage response that would ultimately be lethal to cells. Telomeres, segments of DNA that sit at the ends of chromosomes, resolve this situation by protecting chromosome ends from being mistaken for DNA breaks.
Now, new research shows that two proteins work independently to repress – and control – the activation of damage response pathways that might otherwise cause chromosomes to be harmed.
In research published this week in Nature, Titia de Lange, head of the Laboratory of Cell Biology and Genetics at Rockefeller University, and Eros Lazzerini Denchi, a postdoc in her lab, showed that the two proteins, TRF2 and POT1, independently repress the activation of two DNA damage response pathways at chromosome ends, and further showed how signaling from these pathways, in turn, play a crucial role in DNA repair. The two proteins are part of a protein complex called shelterin, which binds specifically to telomeres and ensures that chromosome ends do not elicit a DNA damage response.
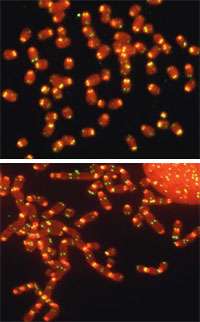
When there is a break in a chromosome or when telomeres shorten, the cell activates a response and stops dividing, reallocating its resources to repair the DNA: Proteins called DNA damage factors are recruited to the site and form foci that can be observed in the microscope. One of these pathways, ATM, looks for double-stranded breaks while the second, ATR, looks for single-stranded DNA, which can form where one strand has begun to degrade.
These series of events can happen anywhere on the chromosome, but Lazzerini Denchi and de Lange found that the shelterin complex has a built-in mechanism to initiate these DNA damage response pathways specifically at telomeres. When Lazzerini Denchi and de Lange deleted TRF2 from the shelterin complex, they saw that this deletion elicited the formation of DNA damage foci at telomeres, indicating that the cell was summoning its resources to repair the damaged telomeres.
However, when they deleted TRF2 in cells that lacked ATM kinase, the primary signaling factor of the pathway, they didn’t see any response. “This really surprised us,” says de Lange. “We thought that both pathways would be activated, but since we didn’t see a damage response after removing both ATM kinase and TRF2, that meant that the ATR pathway wasn’t activated. So something else was controlling the ATR pathway.”
Since POT1 is the only protein within the shelterin complex that binds to single-stranded DNA and single-stranded DNA activates the ATR pathway, de Lange and Lazzerini Denchi thought POT1 was a good candidate. When they tested their theory by deleting POT1, the ATR pathway elicited the formation of DNA damage response foci, a response that was significantly reduced when ATR - but not ATM kinase - levels were reduced in the cell.
The researchers could therefore conclude that POT1 represses ATR and TRF2 represses ATM. “It’s really quite elegant,” says de Lange. “The telomere has been able to create this protein complex – shelterin – that contains two proteins that control the two main pathways that detect and repair DNA damage.”
Lazzerini Denchi and de Lange also found that these two pathways play a crucial role in DNA repair via a reaction called nonhomologous end joining, which links DNA ends without any regard for the sequence at the end. Their work with TRF2 and POT1 showed that either ATM or ATR can stimulate this type of repair.
“We found that DNA repair strongly depends on signaling from these DNA damage response pathways,” says de Lange. “And the only way we could have figured this out is by using telomeres.”
Source: Rockefeller University