Hairpins for Switches: Artificial RNA ligands differentiate between on and off states of riboswitches
How does an organism know when it must produce a protein and in what amount? Clever control mechanisms are responsible for the regulation of protein biosynthesis. One such type of mechanism, discovered only a few years ago, is riboswitches, which function as a sort of “off” switch for the production of certain proteins. These could be a useful point of attack for novel antibiotics if it were possible to find drugs that bind to the switches of pathogens and “turn off” the biosynthesis of essential proteins in bacteria or fungi.
A team at the interdisciplinary Life and Medical Sciences Center at the University of Bonn has now taken a meaningful step toward a better understanding of riboswitches. Researchers led by Michael Famulok have successfully produced hairpin-shaped RNA molecules that are able to differentiate between riboswitches in the on and off states.
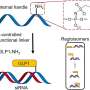
In order to produce a specific protein, a cell first generates a copy of the corresponding gene of the DNA. This blueprint containing the construction plans for the protein is called messenger RNA (mRNA). By using its ribosomes, the cell then reads the mRNA code and synthesizes the protein. Some proteins can activate a “switch” to halt their own synthesis once they are present in sufficient quantity. This is because the mRNA does not only contain the genetic code for the protein but can also contain segments with a switching function.
The protein or a closely connected metabolite binds to this riboswitch and changes its spatial structure such that the mRNA segments controlling the protein production can no longer be read off. For example, when the metabolite thiamine pyrophosphate (TPP) binds to the thiM riboswitch of E. coli bacteria, an mRNA segment recognized by the ribosome as the starting point for “reading” the plan is covered up.
Michael Famulok and his team searched for a probe that can differentiate between off and on. Aptamers are known for their ability to differentiate between different states of proteins. Aptamers are short RNA strands that adopt a specific spatial structure and, like antibodies, selectively bind to specific target molecules. So, why not riboswitches? Over several steps starting from a “library”, a randomly generated large number of highly varied RNA sequences, the scientists selected two short hairpin-shaped aptamers that bind very strongly and specifically to the riboswitch in the “on” position. It turned out that the two hairpins bind to different locations: one to the TPP binding site and the other to a domain responsible for the change in structure of the riboswitch. Both hairpins are crowded when TPP molecules move the riboswitch to the “off” conformation.
Famulok and his team hope to use these aptamers to gain new insights into the function of riboswitches. This could help in the search for a completely new class of antimicrobial agents that block the bacterial thiM riboswitch just like TPP.
Citation: Michael Famulok et al., RNA Ligands That Distinguish Metabolite-Induced Conformations in the TPP Riboswitch, Angewandte Chemie International Edition, doi: 10.1002/anie.200603166
Source: Angewandte Chemie