UO-ONAMI researcher gets patent for nanoparticle-based electronic devices
First came the patent on a greener, faster way to synthesize gold nanoparticles. On Tuesday (March 29), the University of Oregon received a second patent that could lead to a new class of nanoscale electronics and optics assembled from nanoparticles-including ultrasmall transistors that operate efficiently at room temperature.
An article describing the discovery leading to this latest patent was published in the June 2004 issue of Langmuir, the American Chemical Society's surface science journal. The process was developed by UO chemist Jim Hutchison with two of his students, Gerd H. Woehrle and Marvin G. Warner.
"This has been a prolonged effort," Hutchison says. "We reported this invention in 1997 and the original work began in 1996. We had a concept, we reduced it to practice and now we've received a patent on it. It's exciting to have all that effort pay off."
Hutchison's first patent was issued in May 2004. Since then, he says, interest in greener methods for nanotechnology has heated up significantly. At the same time, the quest to build ever-smaller computer chips continues-and Hutchison's new patent may be just the ticket to meet what seems like an endlessly, exponentially increasing demand.
"The first patent covered the use of greener methods to make building blocks. This one is about a greener approach to creating self-assembling structures with those building blocks-a bottom-up approach like using Legos as opposed to chiseling or etching away material like we currently do on silicon chips," Hutchison explains.
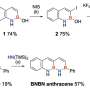
Nanoscale transistors such as those addressed in the patent are composed of nanoparticle building blocks (for example, a chemically functionalized gold core 1.5 nanometers in diameter) and function based upon a mix of classical and quantum mechanical properties.
The patent covers the assembly of devices using a biopolymer DNA as a template. Within living organisms, DNA comprises the genetic code, but by itself, DNA is just a polymer-a string of molecules hooked together in a chain whose links can encode information. In Hutchison's lab, the DNA polymer serves as an architectural scaffold for tiny particles of gold, the ultimate conductor of electricity.
"If you think about a structure of gold dots on a DNA strand, it's like a wire with a whole bunch of minute cuts in it, about 15 angstroms in size," Hutchison says. "In order for electrons to travel down a nanoparticle chain, they have to jump or tunnel from one particle to the next. As a result, these nanochains have different properties than a wire would have. That's why you can make transistors out of them."
This tunneling behavior is a feature of quantum physics that creates problems when using current manufacturing techniques but becomes a boon when the workplace shrinks to nanoscale.
Hutchison directs the UO's Materials Science Institute and is a member of ONAMI, the Oregon Nanoscience and Microtechnologies Institute. The National Science Foundation, the Alfred P. Sloan Foundation and the Camille and Henry Dreyfus Foundation, Inc., have funded his research.
Already known as a world leader for teaching green chemistry principles, Hutchison and his UO colleagues are pioneering the field of green nanoscience. His role in "laying out "the conceptual template for how to design green chemistry nanosubstances" is described in the March issue of Environmental Science & Technology magazine.
Hutchison, 42, is an Oregon native who earned his bachelor's degree at the UO and his doctorate from Stanford. He received an NSF Postdoctoral Fellowship to work on analytical and surface chemistry at the University of North Carolina, Chapel Hill. Since joining the UO faculty in 1994, he has received several awards and honors including an NSF CAREER Award and an Alfred P. Sloan Research Fellowship.
ONAMI is focused on research and commercialization of nanoscience and microtechnologies to foster the creation of new products, companies and jobs in the Pacific Northwest. It unites the University of Oregon (Eugene), Oregon State University (Corvallis), and Portland State University with the Pacific Northwest National Laboratory (Richland, Wash.), the state of Oregon and private industry.
The new patent is No. 6,872,971.
Environmental Science & Technology article (March 2005): pubs.acs.org/subscribe/journal … html/030105tech.html
Source: University of Oregon