Scientists discover novel way to remove iron from ferritin
A new study led by Children's Hospital Oakland Research Institute senior scientist, Elizabeth Theil, Ph.D., is the first to suggest that a small protein or heptapeptide (seven amino acids wrapped into one unit) could be used to accelerate the removal of iron from ferritin. The results of this study may help scientists develop new medications that dramatically improve the removal of excess iron in patients diagnosed with blood diseases such as B-Thalassemia (Cooley's anemia) or Sickle Cell Disease.
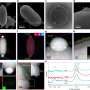
The study appears in this month's issue of the Journal of Biological Chemistry and was conducted by Dr. Theil and her co-authors Xiaofeng S. Liu, postdoctoral fellow at Children's Hospital Oakland Research Institute, Marvin J. Miller, Ph.D. and Leslie D. Patterson, a predoctoral student, both from the University of Notre Dame. The scientists knew that the ferritin protein cage had pores that could open and close. It was also known that chelators (a method to detoxify blood) removed iron faster when the pores were open.
"We wanted to prove a hypothesis that a small protein or peptide could bind to ferritin and could be used to regulate ferritin pores," said Dr. Theil. "Our hypothesis was correct. We proved that when a binding peptide of seven amino acids, a heptapeptide, is coupled with Desferal the rate of removal of iron from ferritin is eight times faster." Desferal is currently used to detoxify the blood of patients with iron overload and is a common therapeutic remedy.
Ferritin is a protein that concentrates iron in its inner core or 'cage'. It plays a critical role in understanding iron overload, which can lead to a variety of symptoms including chronic fatigue, weakness, joint pain and arthritis. If left untreated, iron overload can lead to serious problems, including diabetes, liver and heart disease.
Source: Children's Hospital & Research Center at Oakland