First Detailed View of Molecular Structure May Usher in New Class of Cancer Drugs
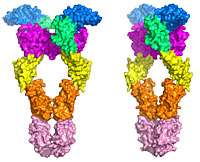
High resolution views of a receptor molecule that is implicated in cancer offer a clear target for the development of a new class of cancer drugs, Yale School of Medicine researchers report July 27 in Cell.
It is also anticipated that the new family of drugs may be applied for the treatment of gastrointestinal stromal tumor (GIST) cancers that are resistant to Gleevec and Sutent. Although these drugs would target the same receptors as Gleevec and Sutent, they would do so by a different mechanism and may therefore be useful for patients who are resistant to these drugs.
“I was surprised to see what the molecules did when they were activated,” said Joseph Schlessinger, professor and chair of pharmacology and senior author of the study. “The arrangement is much simpler and more elegant than I thought.”
The paper, accompanying images, and animation—which looks like a beautiful dance—provide the first detailed, atomic level view of the receptor tyrosine kinase (RTK) Kit before and after it is activated. There are 59 RTK proteins in humans and they play critical roles in the control of a great variety of normal cellular processes as well as in several cancers.
Schlessinger said the newly-revealed mechanism for RTK activation is utilized by many RTKs. “Moreover, these structures offer unexpected new opportunities for drug discovery for the treatment of cancers and other diseases driven by activated RTKs,” he said.
The finding is the result of many years of work involving protein expression, biochemical and biophysical studies and X-ray crystallography, a method used for viewing molecules at a resolution high enough to see atomic detail.
The receptor tyrosine kinase Kit and its natural ligand stem cell factor play an important role in normal blood cell production, gut function, pigmentation, and reproduction. Humans in whom this function is suppressed, or the function is lost, have what is known as a piebald trait—a wing-like discoloration on the chest and abdomen, a white forelock of hair, deafness, and constipation. Mice with the same mutation also have depigmentation and are often anemic and sterile. However, mutations which cause overactivation of Kit are found in a variety of human cancers, among them gastro-intestinal stromal tumors (GIST) and several forms of leukemia.
The structural analysis clearly shows a conformational change as stem cell factor brings two Kit molecules together, resulting in a large rearrangement of and association associations between small parts of the molecules in each pair. Schlessinger said the next step is to identify drugs that can form a wedge in the Kit molecule, making it impossible for them to join together and be activated.
Co-authors include Satoru Yuzawa, Yarden Opatowsky, Zhontang Zhang, Valsan Mandiyan, and Irit Lax.
Source: Yale University
















