Chemists create 'Superbowl' molecule; May lead to better health
In a development that could one day score a touchdown for better health, chemists in Australia have created a 'superbowl' molecule that shows promise for precision drug delivery, according to a recent study in the Journal of the American Chemical Society. Shaped like a miniature football stadium, the molecule is capable of delivering a wide range of drugs — from painkillers to chemotherapy cocktails — to specific areas of the body, potentially resulting in improved treatment outcomes and perhaps saving lives, the researchers say.
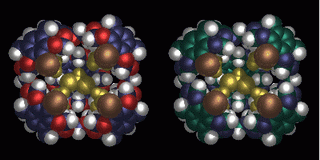
The images above show a three-dimensional molecular model of the superbowl molecule. The molecule, which appears to form a huddle, shows promise of carrying painkillers and chemotherapy drugs to where they are needed in the body. Chemists predict that the molecule will one day improve medical treatments and possibly save lives. (Image courtesy of the American Chemical Society and Australian National University)
The molecule also shows promise for a wide range of other applications, including the removal of environmental toxins and aiding in chemical purification procedures, the investigators say. Their study was published in the Dec. 29 print issue of the journal, one of the peer-reviewed publications of the American Chemical Society, the world's largest scientific society.
The superbowl molecule belongs to a class of artificial bowl-shaped molecules that were first developed in the 1980s to mimic naturally occurring enzymes, which facilitate molecular synthesis and transport within the body. Over the years, these bowl-shaped molecules have been extensively studied and refined by chemists for their wide range of potential uses, including drug delivery, chemical synthesis and as models for cell and viral behavior. Until now, these molecules have had a very limited carrying capacity.
"The compounds we made are bigger versions of these original bowl molecules," says study leader Michael Sherburn, Ph.D., a chemist with Australian National University in Canberra. "These new molecules have much greater capacity and selectivity than their predecessors and show more promise for wider applications."
"I wanted a name for our compounds which acknowledged this and recognized their larger size and shape," says Sherburn. "'Superbowl' was perfect since it conjures up – for me at least – the image of a sports stadium, which the molecules are similar to in shape."
Appropriately, the researcher is also a football fan and watches the Super Bowl each year on Australian television. Super Bowl XXXIX will be played at ALLTEL Stadium in Jacksonville, Fla., on Feb. 6.
The superbowl molecule consists of five concave surfaces, or bowls: four on the outside and one on the bottom. It has an open top, much like an open-air stadium. Its general molecular formula is C268H320O52. The tiny molecules can be modified to include other functional groups that give it additional properties, Sherburn says.
For instance, one highly active version of the superbowl molecule goes by the name tetrabromo-superbowl (C268H320O52Br4) due to the addition of four bromine atoms. The bromine atoms make the hole at the top of the molecule smaller, which makes it more difficult for things to pass in and out. This is ideal if you're trying to capture certain molecules and hold them until they're ready for release, the researcher says.
Superbowl molecules are similar in function to buckyballs, the experimental soccer-ball shaped molecules also known as fullerenes (C60), which also show promise for drug delivery and other useful applications. Whereas a single buckyball is designed to hold either a single atom or a hydrogen molecule, a superbowl molecule can hold large molecules of up to a hundred atoms, particularly those of medicinal value, Sherburn says.
"I'm sure that we will be able to encapsulate compounds such as [the anticancer drug] taxol in the near future," he says, but cautions that the superbowl molecule has not yet been tested in animals or humans. Until then, no one knows yet when this experimental molecule will actually enter the consumer market, but any useful application is likely to take five to ten years, the researcher estimates. Administration of the molecule for drug delivery could be by mouth or by injection, he adds.
There are hurdles to overcome. The current version of the superbowl molecule lacks solubility in water, which could compromise its potential medical applications, the researcher says. Superbowl molecules also have the potential to be toxic. But Sherburn and his associates are confident these potential problems can be solved with further testing and some clever chemical engineering.
Funding for this study was provided by The Australian National University. In addition to Sherburn, study authors include Elizabeth S. Barrett and Alison J. Edwards, also with Australian National University; and Jacob L. Irwin, of the University of Sydney.
The American Chemical Society is a nonprofit organization, chartered by the U.S. Congress, with a multidisciplinary membership of more than 159,000 chemists and chemical engineers. It publishes numerous scientific journals and databases, convenes major research conferences and provides educational, science policy and career programs in chemistry. Its main offices are in Washington, D.C., and Columbus, Ohio.
Source: American Chemical Society (Mark T. Sampson)