January 4, 2006 feature
Faster Nanochannels Water Transport with Sharp Corners
A new study has revealed how sharp corners in nanochannels can speed up the drying rate of liquid water and in turn, transport water faster. "As far as we know, nobody else has realized this gain in water-transport speed," said Dr. Jan Eijkel, from the University of Twente, The Netherlands. Eijkel and his colleagues will publish the findings of this study in a forthcoming issue of Physical Review Letters.
The scientists made sets of parallel channels that were open on both sides. They covered a thin plate of Pyrex glass with a thin layer of photoresist. A mask with the channels outlined on it topped the photoresist and UV light hardened it.
"We removed the photoresist only where the channels would be," Eijkel said. He and his team etched the channels into the glass with hydrofluoric acid and then removed the rest of the photoresist. They then covered the channels with another Pyrex glass plate and bonded the plates at 600 degrees C in preparation for the experiment.
The channels were 4mm long and 72nm high, with widths varying from 2um to 30um. Because of their dimensions, these nanochannels were flat, or had a high aspect ratio. Each nanochannel was an isosceles trapezoid, i.e. non-cylindrical. "Cylindrical channels will give no corner flow and we needed sharp corners," Eijkel said.
First, Eijkel and his colleagues added a drop of demineralized water in one opening of every channel, leading to the channels filling up by capillarity. "A capillarity, such as a nanochannel, will pull in water because its walls like to become wet," Eijkel explained.
Next, the scientists put the nanochannels in a relative humidity chamber until they stabilized. Then, after removing the excess in the nanochannels, the team started the drying process.
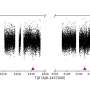
The scientists were then able to view the process by way of the meniscus, the interface between liquid water and air, usually curving towards the glass wall.. "At first, the meniscus was at the exits because the water filled the entire channels. Later, the meniscus moved backwards to the channels’ centers," said Eijkel.
Liquid water had moved to the channels’ centers which is the more humid area. It had also reached the corners instead of the channel exit, which was drier due to evaporation. "Water moved from the corners towards the exit to replenish the evaporating water," Eijkel said. This movement is known as corner flow.
The experiments also included two other fluxes, or methods of water transportation, including film flow and vapor diffusion. Vapor diffusion is the usual way for water to evaporate. A gradient in vapor pressure that borders wet and drier volumes of gas bumps more molecules from wet to dry areas than the other way round.
However, another way to transport water is by film flow. "Water moves within a thin film of absorbed water, going from a place where the film is thick to where it's thinner," Eijkel explained.
Eijkel’s team allowed the three fluxes to be active at the same time. "The water molecules were free to go from the vapor phase to corner flow and back to vapor diffusion," he said.
The scientists noticed that the temperature, which they did not control, varied from 18.5 degrees C to 23.7 degrees C. However, the temperature stayed steady to within + -0.5 degrees C.
The scientists disregarded 15 experiments because the meniscus was on one side or because of dry areas. "Measuring the length of dry channel parts became virtually impossible when dry bits were everywhere," Eijkel clarified.
Eijkel and his team completed 31 experiments, each on a different channel and with a different relative humidity. Corner flow dominated the drying process in 13 experiments.
"Nanochannels dried up to 1000 times faster than you would expect with vapor diffusion alone," said Eijkel. In addition, the drying rate of corner flow did not depend on relative humidity, up to a high humidity of 93%.
The scientists observed that wider channels had more total water volume than narrower channels. "30 micrometer-wide channels held 15 times as much water as 2 micrometer-wide channels. However, the corner flow in narrower channels led to a faster drying rate: the 2 micrometer-wide channels dried 15 times faster than 30 micrometer-wide channels. So, with both channels having two sharp corners, the amount of liquid water transported by each was equal," Eijkel said.
The drying rate in corner flow was inversely proportional to channel width. Vapor diffusion and film flow however, are not dependent on channel width
Each channel in the experiments had two sharp corners of 7 degrees along the 4mm length leaving the remaining two corners blunt. "An enclosed shape with four corners must have a sum equal to 360 degrees, so the other two corners were 173 degrees if they were equal," said Eijkel. The two blunt corners, having very little water, didn't contribute to corner flow.
Another of the scientists’ findings involved the angles of the sharp corners. The amount of water transported through corners increased with the sharpness of the angle. For example, nine times as much water flows through a 60 degree corner,as through one of 90 degrees. The team also experimented with smaller water contact angles, averaging 34 degrees, in order to see the corners hold more water.
Eijkel also believes the findings explain the behaviour of water droplets in drinking vessels of varying levels of cleanliness. "Take water on beer glasses: when the glass is fatty, water runs off in droplets; if the glass is clean, water forms a film. The cleaner or wetter the glass, the smaller the contact area between it and the water droplets," he said.
Eijkel’s research of fast water transport through sharp corners may be useful in the food, paper, textile, and other industries. "Microheat pipes often use corners of 70 degrees, which are sub-optimal," Eijkel noticed. Experimenting with sharp corners may make microheat pipes more efficient.
Copyright 2005 PhysOrg.com, By Syeda Z. Hamdani