Titanium dioxide -- It slices, it dices ...
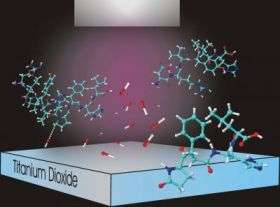
Chemists from the National Institute of Standards and Technology and Arizona State University have proposed an elegantly simple technique for cleaving proteins into convenient pieces for analysis. The prototype sample preparation method, detailed recently in Analytical Chemistry, uses ultraviolet light and titanium dioxide and could be ideal for new microfluidic “lab-on-a-chip” devices designed to rapidly analyze minute amount of biological samples.
Because most proteins are very large, complex molecules made up of hundreds or thousands of amino acids, they usually must be cut up into more manageable pieces for analysis. Today, this most commonly is done by using special enzymes called “proteases” that sever the chains at well-known locations. The protease trypsin, for example, cuts proteins at the locations of the amino acids lysine and arginine.
Analyzing the residual fragments can identify the original protein. But enzymes are notoriously fussy, demanding fairly tight control of temperature and acidity, and the enzymatic cutting process can be time-consuming, from a matter of hours to days.
For a “radically” different approach, the NIST group turned to a semiconductor material, titanium dioxide. Titanium dioxide is a photocatalyst—when exposed to ultraviolet light its surface becomes highly oxidizing, converting nearby water molecules into hydroxyl radicals, a short-lived, highly reactive chemical species. In the NIST experiments, titanium dioxide coatings were applied to a variety of typical microanalysis devices, including microfluidic channels and silica beads in a microflow reactor. Shining a strong UV light on the area, in the presence of a protein solution, creates a small “cleavage zone” of hydroxyl radicals that rapidly cut nearby proteins at the locations of the amino acid proline.
Although development work remains to be done, according to the researchers, the NIST photocatalysis technique offers several advantages over conventional enzyme cleavage of proteins. It’s not particularly sensitive to temperature or acidity, and needs no additional reagents other than dissolved oxygen in the solution. It’s a simple arrangement, easy to incorporate into a wide range of instruments and devices, and titanium dioxide, an inorganic material, will last virtually forever in a broad range of conditions—enzymes have to be treated carefully and stored in temperature-controlled environments. The target amino acid, proline, is relatively sparse in most proteins, but it’s found at key locations, such as sharp turns in the molecule, that aid analysis. And it’s fast—in trials with the protein angiotensin I, the team obtained detectable cleavage patterns in as little as 10 seconds.
Citation: B.J. Jones, M.J. Vergne, D.M. Bunk, L.E. Locascio and M.A. Hayes. Cleavage of peptides and proteins using light-generated radicals from titanium dioxide. Anal. Chem. 2007, 79, 1327-1332.
Source: National Institute of Standards and Technology



















